Overview
Transcranial magnetic stimulation (TMS) is a procedure that uses magnetic fields to stimulate nerve cells in the brain to improve symptoms of major depression. It's called a "noninvasive" procedure because it's done without using surgery or cutting the skin. Approved by the U.S. Food and Drug Administration (FDA), TMS usually is used only when other depression treatments haven't been effective.
The FDA also approved TMS for obsessive-compulsive disorder (OCD), migraines and to help people stop smoking when standard treatments haven't worked well. Research continues into other potential uses for TMS, including epilepsy.
When used for depression, OCD and to stop smoking, this treatment involves delivering repeated magnetic pulses, so it's called repetitive TMS or rTMS.
How it works
Repetitive transcranial magnetic stimulation (rTMS)
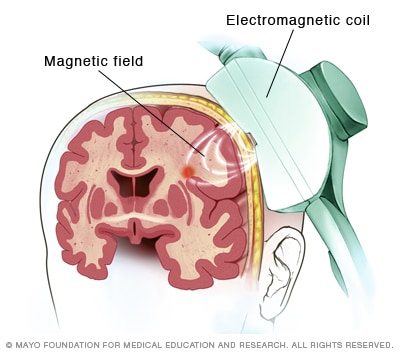
Repetitive transcranial magnetic stimulation (rTMS)
In repetitive transcranial magnetic stimulation (rTMS), an electromagnetic coil placed against the scalp creates a magnetic field that stimulates certain areas of the brain.
During an rTMS session for depression, an electromagnetic coil is placed against the scalp of your head. This coil delivers magnetic pulses that stimulate nerve cells in the region of your brain involved in mood control and depression. It's thought to activate regions of the brain that have decreased activity during depression.
You also may hear the term "deep transcranial magnetic stimulation" or "deep TMS." The difference between rTMS and deep TMS has to do with the type of coil used. The deep TMS coil stimulates deeper and wider areas of the brain, compared with rTMS. Deep TMS coils have been FDA-approved for OCD and to stop smoking.
Although the biology of why TMS works isn't completely understood, the stimulation appears to affect how the brain is working. It seems to ease depression symptoms and improve mood.
There are different ways to perform TMS. Methods may change as experts learn more about the most effective ways to perform treatments.
Products & Services
Why it's done
Depression is a treatable condition. But for some people, standard treatments aren't effective. Repetitive TMS may be used when standard treatments such as medicines, and talk therapy, known as psychotherapy, don't work.
TMS is sometimes used to treat OCD, migraines and to help people stop smoking after other treatments have not been successful.
Risks
Repetitive TMS is a noninvasive form of brain stimulation. Unlike vagus nerve stimulation or deep brain stimulation, rTMS does not require surgery or implanting electrodes. And, unlike electroconvulsive therapy (ECT), rTMS doesn't cause seizures or memory loss. It also doesn't require the use of anesthesia, which puts people in a sleep-like state.
Generally, rTMS is considered safe and well-tolerated. However, it can cause some side effects.
Common side effects
Side effects are generally mild to moderate and improve shortly after a session. Side effects decrease over time with more sessions.
Side effects may include:
- Scalp discomfort and pain.
- Headache.
- Tingling, spasms or twitching of facial muscles.
- Lightheadedness.
Your health care provider can adjust the level of stimulation to reduce symptoms. Or your provider may recommend that you take a pain medicine available without a prescription before the procedure. In some people who get frequent headaches or migraines, TMS triggers headaches, so treatments might need to be stopped.
Uncommon side effects
Serious side effects are rare but may include:
- Seizures.
- Emotional highs, called mania, particularly in people with bipolar disorder.
- Hearing loss if the ears aren't well-protected during treatment.
More study is needed to determine whether rTMS may have long-term side effects.
How you prepare
Before having rTMS, you may need a:
- Physical exam and possibly lab tests or other tests.
- Mental health evaluation to discuss your depression.
These evaluations help make sure that rTMS is a safe option for you.
Tell your health care provider if:
- You're pregnant or thinking of becoming pregnant.
- You have metal or implanted medical devices in your body. In some cases, people with metal implants or devices can have rTMS. But due to the strong magnetic field produced during rTMS, it's not recommended for some people who have these devices:
- Aneurysm clips or coils.
- Stents.
- Implanted stimulators.
- Implanted vagus nerve or deep brain stimulators.
- Implanted electrical devices, such as pacemakers or medicine pumps.
- Electrodes for monitoring brain activity.
- Cochlear implants for hearing.
- Magnetic implants.
- Bullet fragments.
- Other metal devices or objects implanted in their body.
- You're taking medicines, including prescriptions, medicines available without a prescription, herbal supplements, vitamins or other supplements, and the doses.
- You have a history of seizures or a family history of epilepsy.
- You have other mental health conditions, such as issues with alcohol or drugs, bipolar disorder, or psychosis.
- You have brain damage from illness or injury, such as a brain tumor, stroke or traumatic brain injury.
- You have frequent or severe headaches.
- You have any other medical conditions.
- You've had treatment with rTMS in the past and whether it was helpful in treating your depression.
Before your first appointment
Repetitive TMS isn't invasive, doesn't require anesthesia and can be performed on an outpatient basis. While you don't need to arrange for someone to drive you home after treatment, you may prefer to be driven home after the first session to get a sense of how you'll feel afterward.
Before considering treatment, check with your health insurance company to see whether rTMS is covered. Most policies cover rTMS for depression but require certain medical necessity conditions to be met. These conditions include trying a minimum number of antidepressants that have not worked. Repetitive TMS for OCD and for stopping smoking is new enough that most insurance policies don't cover its use for these issues. That may change as its use becomes more widespread.
What you can expect
Repetitive TMS is usually done in a health care provider's office or clinic. It requires a series of treatment sessions to be effective. Generally, sessions are carried out daily, five times a week, for 4 to 6 weeks.
Your first treatment
Before treatment begins, your health care provider will need to identify the best place to put the magnet on your head and the best dose of magnetic energy for you. Your first appointment usually lasts about an hour.
During your first appointment, the "mapping" process is performed. Here's what you can likely expect:
- You'll be taken to a treatment room, asked to sit in a reclining chair and given earplugs to wear during the procedure.
- An electromagnetic coil will be placed against your head and switched off and on repeatedly to produce stimulating pulses. This results in a tapping sensation on your head and a clicking sound, followed by a pause.
- The coil will be moved around your head and the magnetic energy adjusted to find the location that causes the fingers or hands on the other side of your body to twitch. Once the location is found, a second process of finding the "motor threshold" occurs. Strong and weak pulses alternate to determine how much energy is needed to move your fingers or thumb during at least half of the attempts.
During each treatment
The coil placement and dose are usually done only once. This occurs during the first treatment. For all later treatments, the magnet is moved to the treatment position using measurements from the first treatment.
Here's what to expect during each treatment:
- You'll sit in a comfortable chair, wearing earplugs, with the magnetic coil placed against your head. The treatment location of the coil is different from where the coil makes your fingers or thumb move.
- When the machine is turned on, you'll feel and hear rapid tapping on your scalp. The pattern will be a few seconds of tapping followed by a pause. The pattern will repeat. You may have scalp discomfort and even some pain during the tapping, but not during the pauses. You'll remain awake and alert during the procedure.
- Depending on the type of stimulation pattern used, the procedure will last 3.5 minutes or 20 minutes. The latest stimulation type is called "intermittent theta burst stimulation" and requires only 3.5 minutes. The original rTMS pattern took 37 minutes, but it can be performed in 20 minutes now.
After each treatment
You can return to your normal daily activities after your treatment. You might have a headache for a short time afterward. Between treatments, you can usually expect to work and drive.
Results
If rTMS works for you, your depression symptoms may improve or go away completely. Symptom relief may take a few weeks of treatment.
The effectiveness of rTMS may improve as researchers learn more about techniques, the number of stimulations needed and the best sites on the brain to stimulate.
Ongoing treatment
After completing an rTMS treatment series for depression, standard care — such as medicine and talk therapy — may be recommended as ongoing treatment.
It's not yet known if maintenance rTMS sessions will benefit your depression. This involves continuing treatment when you are symptom-free with the hope that it will prevent the return of symptoms.
If your depression improves with rTMS for at least 2 to 3 months and then you have symptoms later, you can repeat rTMS treatment. Most insurance companies will cover repeat treatment.
Clinical trials
Explore Mayo Clinic studies of tests and procedures to help prevent, detect, treat or manage conditions.