Overview
Implantable cardioverter-defibrillator (ICD)
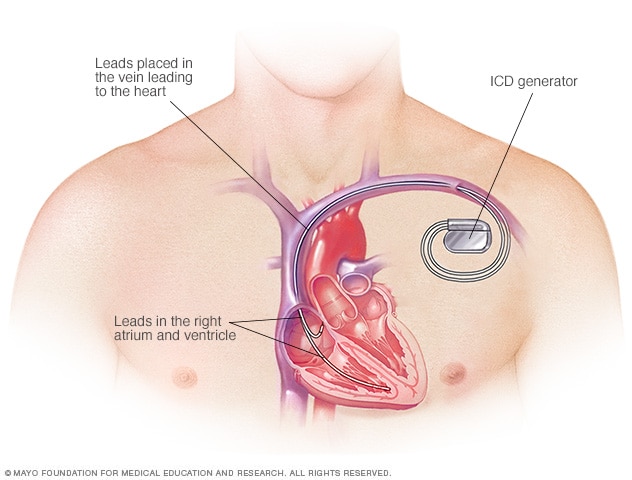
Implantable cardioverter-defibrillator (ICD)
An ICD controls the heartbeat by delivering shocks to the heart when the device detects an irregular heartbeat.
An implantable cardioverter-defibrillator, also called an ICD, is a small battery-powered device placed in the chest. It detects and stops irregular heartbeats, also called arrhythmias. An ICD constantly checks the heartbeat. It delivers electric shocks, when needed, to restore a regular heartbeat.
You might need an ICD if you have a dangerously fast heartbeat called ventricular tachycardia or ventricular fibrillation. An ICD also may be recommended if you're at high risk of dangerous irregular heartbeats, usually because of a weak heart muscle.
An ICD isn't the same as a pacemaker. A pacemaker is a device used to prevent dangerously slow heartbeats.
Types
An ICD is a type of cardiac therapy device. There are two basic types:
- A traditional ICD is placed in the chest. Wires, called leads, attach to the heart.
- A subcutaneous ICD, also called an S-ICD, is placed under the skin at the side of the chest below the armpit. It's attached to a sensor, called an electrode. The sensor runs along the breastbone. An S-ICD is larger than a traditional ICD. It doesn't touch the heart.
Products & Services
Why it's done
Subcutaneous implantable cardioverter-defibrillator (S-ICD)
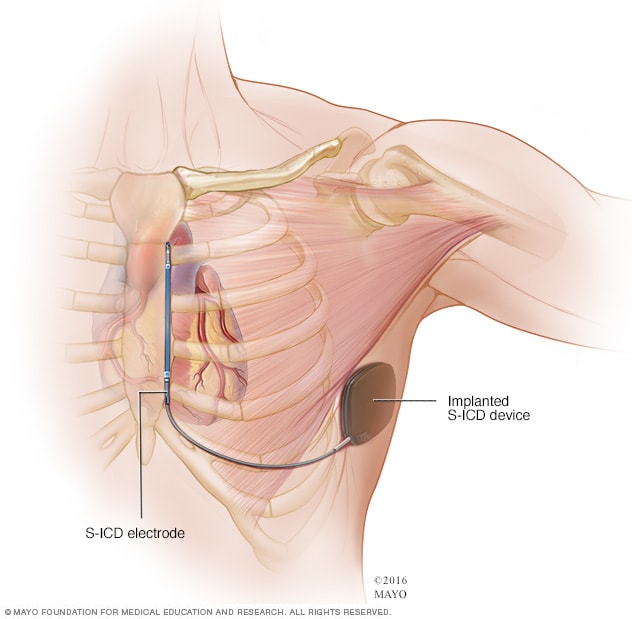
Subcutaneous implantable cardioverter-defibrillator (S-ICD)
A subcutaneous implantable cardioverter-defibrillator (S-ICD) is a less invasive alternative to a traditional ICD. The S-ICD device is placed under the skin at the side of the chest below the armpit. It connects it to a sensor that runs along the breastbone.
An ICD constantly checks for irregular heartbeats and instantly tries to correct them. It helps when there is a sudden loss of all heart activity, a condition called cardiac arrest.
An ICD is the main treatment for anyone who has survived cardiac arrest. More and more, the devices are used in people at high risk of sudden cardiac arrest. An ICD lowers the risk of sudden death from cardiac arrest more than medicine alone.
Your heart doctor may recommend an ICD if you have symptoms of an irregular heartbeat called sustained ventricular tachycardia. Fainting is one of the symptoms.
An ICD also may be recommended if you survived cardiac arrest or if you have:
- A history of coronary artery disease and a heart attack that has weakened the heart.
- An enlarged heart muscle.
- A genetic heart condition that raises the risk of dangerously fast heartbeats, such as some types of long QT syndrome.
Risks
Possible risks of implantable cardiac defibrillators or ICD surgery may include:
- Infection at the implant site.
- Swelling, bleeding or bruising.
- Blood vessel damage from ICD wires.
- Bleeding around the heart, which can be life-threatening.
- Blood leaking through the heart valve where the ICD lead is placed.
- Collapsed lung.
- Movement of the device or leads, which could lead to a rip or cut in the heart muscle. This complication, called cardiac perforation, is rare.
How you prepare
Before you get an ICD, some tests are done to check your heart health. Tests may include:
- Electrocardiogram, also called ECG or EKG. An ECG is a quick and painless test that checks the heartbeat. Sticky patches called electrodes are placed on the chest and sometimes the arms and legs. Wires connect the electrodes to a computer, which displays or prints the test results. An ECG can show if the heart is beating too fast or too slowly.
- Echocardiogram. This imaging test uses sound waves to create moving pictures of the heart. It shows the size and structure of the heart and how blood flows through the heart.
-
Holter monitoring. A Holter monitor is a small, wearable device that keeps track of the heartbeat. You usually wear it for 1 to 2 days. A Holter monitor may be able to spot irregular heartbeats that an ECG missed. Wires from sensors that stick to the chest connect to a battery-powered recording device. You carry the device in a pocket or wear it on a belt or shoulder strap.
While wearing the monitor, you may be asked to write down your activities and symptoms. Your healthcare team may compare your notes with the device recordings and try to figure out the cause of your symptoms.
- Event monitor. This portable ECG device is intended to be worn for up to 30 days or until you have an arrhythmia or symptoms. You usually press a button when symptoms occur.
- Electrophysiology study, also called EP study. This test may be done to confirm a diagnosis of a fast heartbeat. It also can find the area in the heart that's causing an irregular heartbeat. A healthcare professional guides a flexible tube called a catheter through a blood vessel into the heart. Often, more than one catheter is used. Sensors on the tip of each catheter record the heart's signals.
What you can expect
Before
If you're getting an ICD, you'll likely be asked not to eat or drink for a few hours before the surgery to place the device.
Tell your healthcare team about all the medicines you take. Ask whether you can take them before the surgery to get an ICD.
Make plans to have someone to drive you home after your ICD surgery. You also need someone to help you while you are recovering.
During
A member of your healthcare team inserts an IV into your forearm or hand. You may get medicine called a sedative to help you relax. Several sticky patches called electrodes are placed on your chest and sometimes the legs. The patches connect to a machine that constantly checks your heartbeat during the surgery.
Sometimes you're awake when getting an ICD. Or you may get a combination of medicines to put you in a sleep-like state. This is called general anesthesia. Your healthcare team explains the type of sedation you'll need.
To place a traditional ICD, the surgeon makes a small cut in the skin in the chest area. One or more flexible wires called leads are inserted into a blood vessel near the collarbone. The surgeon guides the wires to the heart. One end of each wire attaches to the heart. The other ends attach to a device called a shock generator. The device is placed under the skin beneath the collarbone.
The surgery to place an ICD in the chest usually takes a few hours.
Once the ICD is in place, your surgeon tests it and programs it for your specific heart rhythm needs. Testing the ICD might require speeding up the heart and then shocking it back into a regular rhythm.
Depending on the exact issue with your heartbeat, an ICD could be programmed for:
- Low-energy pacing. You may feel nothing or a painless fluttering in your chest when your ICD responds to mild changes in your heartbeat.
- A higher energy shock. For more-serious heart rhythm problems, the ICD may deliver a higher energy shock. This shock can be painful, and it might make you feel as if you've been kicked in the chest. The pain usually lasts only a second. There shouldn't be discomfort after the shock ends.
Usually, only one shock is needed to restore a regular heartbeat. Some people might have two or more shocks during a 24-hour period.
Having three or more shocks in a short amount of time is called an electrical or arrhythmia storm. If you have an electrical storm, you should call 911 or seek emergency medical help. The healthcare team checks to see if your ICD is working properly or if you're having irregular heartbeats.
If needed, the ICD can be programmed again to lessen how many shocks you get or how often you get them. Medicines also may be needed to control the heartbeat and lower the risk of an ICD electrical storm.
After
You'll usually go home on the day after the ICD surgery.
The area where the ICD is placed may be swollen and tender for a few days or weeks. Your healthcare professional might prescribe pain medicine. Do not take aspirin and ibuprofen. They may raise the risk of bleeding.
For about eight weeks after getting an ICD, do not make any sudden movements that raise your left arm above your shoulder. You don't want the device wires to move until the area is healed. You may need to limit your driving, depending on the type of ICD received. Your healthcare team gives you instructions on when it's safe to return to driving and other daily activities.
For about four weeks after getting an ICD, your healthcare team may say not to do the following activities:
- Energetic sports or activities that may lift the left arm above the shoulder. For example, golf, tennis, swimming, bowling or vacuuming.
- Heavy lifting.
- Other types of strenuous exercise.
Your healthcare professional may tell you not to play contact sports after getting an ICD. A hit to the chest area may damage the ICD or cause its wires to move.
Electrical signals from other devices
Problems with an ICD due to electrical signals from other devices or items are rare. Still, take care with the following:
- Cellular phones and other mobile devices. It's safe to talk on a cellphone if you have an ICD. But do not place the cellphone within 6 inches (about 15 centimeters) of an ICD site when the phone is turned on. Even though it's not likely, an ICD could mistake a cellphone's signal for a heartbeat and slow the heartbeat. This might cause symptoms such as sudden fatigue.
- Security systems. After getting an ICD, you'll receive a card that says you have one. Show your card to airport security workers because the ICD may set off security alarms.
- Metal detectors. Hand-held metal detectors, such as those used at airport security checks, often have a magnet that can interfere with an ICD. Do not scan the area over an ICD for more than 30 seconds. At the airport, you can request a manual search instead.
- Medical equipment. Tell all members of your healthcare team that you have an ICD. Imaging tests that use magnetic resonance imaging, also called MRI, may not be recommended if you have an ICD. Treatments that use heat energy, called radiofrequency ablation, also may not be recommended. Your healthcare team can tell you if you need a different test or treatment.
- Power generators. Stand at least 2 feet (0.6 meters) from welding equipment, high-voltage transformers or motor-generator systems. If you work around such equipment, your healthcare team can arrange a test in your workplace to see if the equipment affects your ICD.
- Headphones and wireless chargers. Headphones may contain a magnetic substance that can interfere with an ICD. Keep headphones and wireless chargers at least 6 inches (about 15 centimeters) from an ICD.
- Magnets. Magnets may affect an ICD, so it's a good idea to keep magnets at least 6 inches (15 centimeters) from the ICD site.
Devices that have little or no risk to an ICD include microwave ovens, televisions and remote controls, AM/FM radios, toasters, electric blankets, electric shavers and electric drills, computers, scanners, printers, and GPS devices.
Driving restrictions
If you have an ICD to treat a life-threatening irregular heartbeat called ventricular arrhythmia, driving a vehicle may pose risks to yourself and others. The combination of this heart condition and shocks from an ICD can cause fainting, which would be dangerous while driving.
Many countries have driving restrictions for people with ICDs. If you have an ICD because of cardiac arrest or ventricular arrhythmia, your healthcare professional may tell you to wait several months before driving or operating a vehicle. If you get a shock from the ICD, tell your healthcare team, even if you do not faint. Usually, you'll be told not to drive until you've been shock-free for several months.
If you have an ICD but no history of life-threatening irregular heartbeats, you can usually return to driving about a week after getting an ICD if you've had no shocks. Talk to your healthcare team about your specific restrictions.
You usually can't get a commercial driver's license if you have an ICD.
Results
After getting an ICD, you need regular health appointments to check your heart and the device.
The lithium battery in an ICD can last 5 to 7 years. The battery usually is checked during regular health appointments, which should occur about every six months. Ask your healthcare team how often you need a checkup. When the battery is nearly out of power, the generator is replaced with a new one. This involves getting a minor surgery. Usually, you go home the same day.
Tell your healthcare professional if you have any shocks from your ICD. The shocks might make you worried. But they mean the ICD is treating a heart rhythm condition and protecting against sudden death.
ICDs and end-of-life issues
If you have an ICD and become terminally ill, your ICD still delivers shocks if it isn't turned off. A healthcare professional can perform a simple procedure to turn off the ICD, if desired. Turning off the device can prevent unwanted shocks and needless suffering. Doing so won't cause the heart to stop right away.
Talk to your healthcare team about your wishes. Also talk to family members or the person chosen to make medical decisions for you in an end-of-life care situation.
Clinical trials
Explore Mayo Clinic studies of tests and procedures to help prevent, detect, treat or manage conditions.
Feb. 18, 2025