Overview
Vagus nerve stimulation involves using a device to stimulate the vagus nerve with electrical impulses. There's one vagus nerve on each side of your body. The vagus nerve runs from the lower part of the brain through the neck to the chest and stomach. When the vagus nerve is stimulated, electrical impulses travel to areas of the brain. This alters brain activity to treat certain conditions.
Vagus nerve stimulation can be done in many ways with many devices. An implantable vagus nerve stimulator has been approved by the Food and Drug Administration (FDA) to treat epilepsy and depression. The device works by sending stimulation to areas of the brain that lead to seizures and affect mood.
Another vagus nerve stimulation device is FDA approved to be used with rehabilitation when recovering from a stroke. When someone has trouble using their hand or arm after a stroke, the stimulation helps create new pathways in the brain as the person performs exercises. This can help the person regain function.
These devices are surgically implanted under the skin of the chest. A wire is threaded under the skin connecting the device to the left vagus nerve. When activated, the device causes electrical signals to be sent along the left vagus nerve to the brainstem, which then affects the brain. The right vagus nerve isn't typically used because stimulation is more likely to affect the function of the heart.
There also are newer, noninvasive vagus nerve stimulation devices that don't require surgery. The FDA has approved a noninvasive vagus nerve stimulation device to treat cluster headaches and migraine in the United States. This device stimulates the vagus nerve when it's held against the skin of the neck. The stimulation blocks pain signals to prevent or relieve head pain.
Researchers are studying vagus nerve stimulation as a potential treatment for a variety of other conditions. Current research is looking at the potential benefits of the therapy for rheumatoid arthritis, inflammatory bowel disease, bipolar disorder, obesity and Alzheimer's disease.
Products & Services
Why it's done
Vagus nerve stimulation
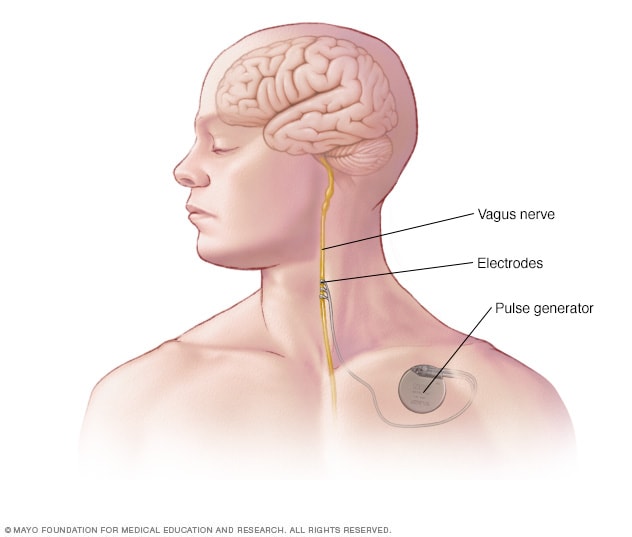
Vagus nerve stimulation
In vagus nerve stimulation, an implanted pulse generator and lead wire stimulate the vagus nerve, which leads to stabilization of irregular electrical activity in the brain.
A variety of conditions can be treated with implantable vagus nerve stimulation devices.
Epilepsy
About one-third of people with epilepsy don't fully respond to anti-seizure drugs. Vagus nerve stimulation may be an option to reduce seizures in people who haven't achieved control with medicines.
The FDA has approved vagus nerve stimulation for people who:
- Are 4 years old and older.
- Have focal epilepsy. In focal epilepsy, the brain activity that causes seizures happens in one area of the brain.
- Have seizures that aren't well-controlled with medicines.
Vagus nerve stimulation also is considered for people with generalized epilepsy.
Depression
Vagus nerve stimulation also may be helpful for people who haven't responded to depression treatments. It may be used when medicines, psychological counseling and electroconvulsive therapy haven't been effective for depression.
The FDA has approved vagus nerve stimulation for the treatment of depression in adults who:
- Have long-lasting, hard-to-treat depression, known as treatment-resistant depression.
- Haven't improved after trying four or more medicines or electroconvulsive therapy (ECT), or both.
- Continue standard depression treatments along with vagus nerve stimulation.
Stroke recovery
For people who are recovering from a stroke, vagus nerve stimulation has been FDA-approved when combined with rehabilitation. Vagus nerve stimulation paired with rehabilitation may help people recover function in their hands and arms after an ischemic stroke.
Risks
Having a vagus nerve stimulator implanted is safe for most people. But it does have some risks, both from the surgery to implant the device and from the brain stimulation.
Surgery risks
Surgical complications with implanted vagus nerve stimulation are rare and are similar to the dangers of having other types of surgery. They include:
- Pain where the cut is made to implant the device.
- Infection.
- Difficulty swallowing.
- Vocal cord paralysis. This is usually temporary but can be permanent.
Side effects after surgery
Some of the side effects and health problems associated with implanted vagus nerve stimulation can include:
- Voice changes.
- A hoarse voice.
- Throat pain.
- Cough.
- Headaches.
- Shortness of breath.
- Trouble swallowing.
- Tingling or prickling of the skin.
- Trouble sleeping.
- Worsening of sleep apnea.
For most people, side effects are tolerable and typically lessen over time. However, some side effects may remain for as long as you use implanted vagus nerve stimulation.
Adjusting the electrical impulses can help minimize these effects. If you can't tolerate the side effects, the device can be shut off.
How you prepare
It's important to carefully consider the pluses and minuses of implanted vagus nerve stimulation before having the procedure. Make sure you know all of your other treatment choices. Be sure that you and your health care provider feel that implanted vagus nerve stimulation is the best option for you. Ask your provider exactly what to expect during surgery and after the pulse generator is in place.
Food and medicines
You may need to stop taking certain medicines ahead of surgery. Your surgeon may ask you not to eat for a certain number of hours before the procedure.
What you can expect
Before the procedure
Before surgery, you'll have a physical exam. Your surgeon may have you start taking antibiotics before surgery to prevent infection.
During the procedure
Surgery to implant the vagus nerve stimulation device often is done on an outpatient basis in which you go home afterward. During surgery, you'll likely be put into a sleep-like state. This is done with general anesthesia.
The surgery itself doesn't involve the brain. Two cuts, known as incisions, are made. One is made on the chest or in the armpit area. The other is made on the left side of the neck.
The pulse generator is implanted in the upper left side of the chest. The pulse generator is about the size of a stopwatch and runs on battery power. A lead wire is connected to the pulse generator. The lead wire is guided under your skin from your chest up to your neck. Then it's attached to the left vagus nerve through the second incision. The device and its battery last for many years before being exchanged for a new device. It also can be removed if necessary.
Video: Vagus nerve stimulation
It’s called vagus nerve stimulation. Surgeons implant a device near the collarbone and run a wire to the vagus nerve. When the device fires it stimulates that nerve to send signals to the brain. This increases activity in areas that control mood.
After the procedure
If you had the device implanted to treat epilepsy or depression, the pulse generator is turned on during a medical visit a few weeks after surgery. Then it can be programmed to deliver electrical impulses to the vagus nerve at various durations, frequencies and currents. Vagus nerve stimulation usually starts at a low level. It's gradually increased depending on your symptoms and side effects.
Stimulation is programmed to turn on and off in cycles — such as 30 seconds on, five minutes off. You may have some tingling sensations or slight pain in your neck. You also may have a hoarse voice when the device is on.
Newer models that treat epilepsy also stimulate the nerve when there's a rapid increase in heart rate, which may indicate a seizure. You can use a hand-held magnet to start stimulation at a different time. For example, you can turn it on if you sense you're about to have a seizure.
If you've had a device implanted after a stroke, the device is typically turned on by a specialist during rehabilitation. You also can turn it on at home using a magnet. You might turn it on when you're doing certain tasks such as cooking.
You'll need to visit your health care provider periodically to make sure that the pulse generator is working correctly and that it hasn't shifted out of position. Check with your health care provider before having any medical tests, such as an MRI. The test might interfere with the device.
Results
If you had the device implanted for epilepsy, it's important to understand that vagus nerve stimulation isn't a cure. Most people with epilepsy won't stop having seizures. They'll also likely continue taking epilepsy medicine after the procedure. But many might have fewer seizures — up to 50% fewer. The seizures also may be less intense.
It can take months or even a year or longer of stimulation before you notice any significant reduction in seizures. Vagus nerve stimulation also may shorten the recovery time after a seizure. People who have had vagus nerve stimulation to treat epilepsy may experience improvements in mood and quality of life.
Research on the benefits of implanted vagus nerve stimulation for the treatment of depression is ongoing. Some studies suggest the benefits of vagus nerve stimulation for depression build over time. It may take at least several months of treatment before you notice any improvements in depression symptoms. Implanted vagus nerve stimulation doesn't work for everybody, and it isn't intended to replace traditional treatments.
Studies have found that vagus nerve stimulation paired with rehabilitation helped improve function in people who had a stroke. It also may help people who have problems with thinking and swallowing after a stroke. Research is ongoing.
Some health insurance carriers may not pay for this procedure.
Studies of implanted vagus nerve stimulation as a treatment for conditions such as Alzheimer's disease, rheumatoid arthritis, inflammatory bowel conditions and heart failure have been too small to draw any definitive conclusions. More research is needed.
Clinical trials
Explore Mayo Clinic studies of tests and procedures to help prevent, detect, treat or manage conditions.
The Mayo Clinic experience and patient stories
Our patients tell us that the quality of their interactions, our attention to detail and the efficiency of their visits mean health care like they've never experienced. See the stories of satisfied Mayo Clinic patients.