Overview
Contraceptive implant

Contraceptive implant
A contraceptive implant works to prevent pregnancy by releasing a low, steady dose of the hormone progestin.
Contraceptive implants are a long-term birth control method. They're also called long-acting reversible contraception, or LARC.
A contraceptive implant is a flexible plastic rod about the size of a matchstick that is placed under the skin of the upper arm. The implant releases a low, steady dose of the hormone progestin.
Progestin prevents pregnancy by pausing ovulation. And it thickens the mucus of the cervix. This makes it hard for sperm to reach an egg. Progestin also thins the lining of the uterus. If sperm do reach an egg, this makes it harder for the fertilized egg to attach to the uterus.
Products & Services
Why it's done
Contraceptive implants are effective, long-term birth control. Benefits of the implant include:
- It's reversible. A care provider can remove the implant anytime you decide it's not right for you or you want to get pregnant.
- You don't have to think about it. You'll need to replace it every three years. But you won't have to worry about it every day or every month like other methods.
- You're in charge of your birth control. No need to pause sex or get your partner to agree to birth control.
- It's estrogen-free. Methods containing estrogen can raise the risk of forming blood clots. So, the implant may be a better choice for you if you want a lower-risk option.
- It allows a quick return to fertility. If you want to get pregnant, you can start trying as soon as the implant is removed.
But contraceptive implants aren't right for everyone. Your care team might suggest another birth control method if you have:
- Allergies to any parts of the implant.
- A history of serious blood clots, heart attack or stroke.
- Liver tumors or disease.
- A history of breast cancer, or if you might have breast cancer.
- Bleeding outside of your typical period that hasn't been checked by a care provider.
The label for the active ingredient in the implant, etonogestrel, says it shouldn't be used by people with a history of blood clots. The warning comes from studies of combination birth control pills that also use progestin plus estrogen. But those risks may be due to the estrogen alone. Because the implant only uses progestin, it isn't clear if it actually carries any risk of forming blood clots.
Talk to your care team if you might be at risk for blood clots. This includes a history of blood clots in your legs or lungs, also called pulmonary embolus. They will know if the implant is a safe method for you.
Also, tell your care team if you have a history of:
- Allergies to anesthetics or antiseptics.
- Depression.
- Diabetes.
- Gallbladder disease.
- High blood pressure.
- High cholesterol or high triglycerides.
- Seizures or epilepsy.
Certain medicines and herbal products may lower the levels of progestin in your blood. This means the implant might not prevent pregnancy as well. Medicines known to do this include certain seizure medicines, sedatives, HIV medicines and the herb St. John's wort. If you take any of these medicines, talk with your care team about your birth control options.
Risks
The contraceptive implant doesn't protect against sexually transmitted infections.
Fewer than 1 in 100 women who use the contraceptive implant for one year will get pregnant. But if you do conceive while using the implant, there's a higher chance the pregnancy will be ectopic. This means the fertilized egg implants outside the uterus, often in a fallopian tube. But the risk of ectopic pregnancy is still lower than for those who are having sex without birth control. That's because the rate of pregnancy while using the implant is so low.
Side effects associated with contraceptive implants include:
- Pain in the back or stomach area.
- Changes to your period. It may stop completely. This is called amenorrhea.
- Higher risk of noncancerous, or benign, ovarian cysts.
- Lower sex drive.
- Dizziness.
- Headaches.
- Mild insulin resistance.
- Mood swings and depression.
- Nausea or upset stomach.
- Possible problems with other medicines.
- Sore breasts.
- Vaginal soreness or dryness.
- Weight gain.
How you prepare
Your care team will look at your whole health before moving forward with scheduling the procedure. If all looks safe, they will decide the best date to place the implant. This is based on your period cycle and any birth control method you're using.
You might have to take a pregnancy test before the implant can be placed. Once the implant is in, it's a good idea to use condoms or another nonhormonal backup method of birth control for the first week just to be safe.
You might not need backup birth control if you have the contraceptive implant placed:
- In the first five days of your period. Even if you're still bleeding or didn't use birth control before.
- In the first seven days of your period after correctly using hormonal birth control such as combination pills, the ring or the patch.
- While taking the minipill every day as prescribed.
- The day your injection is due if you've been using the birth control shot (Depo-Provera).
- The day of or a few days before another contraceptive implant or intrauterine device (IUD) you've used is removed.
What you can expect
Contraceptive implant location
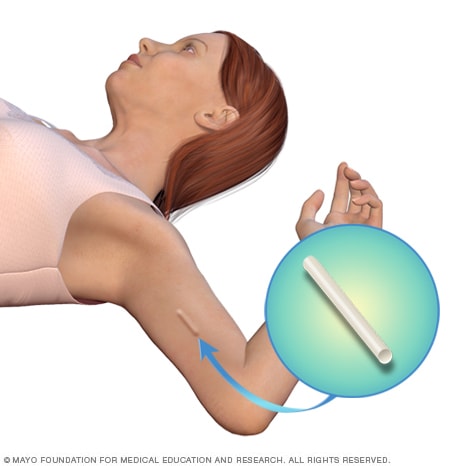
Contraceptive implant location
The contraceptive implant is placed under the skin of the upper arm. You won't be able to see the implant. But you may be able to feel the implant underneath the skin if you touch that spot on your arm.
You'll have the contraceptive implant placed at your care provider's location. The actual procedure takes just a minute or so, though preparation will take a bit longer.
During the procedure
You'll lie on your back and be awake. The arm that will get the implant will either be straight out to the side, or bent at the elbow and placed near your head. These positions show the inside of your upper arm.
Your provider will find a groove between the muscles on the inside of your upper arm. Then, you'll get a shot of local anesthetic to numb the area. Last, the device will be inserted with an applicator just beneath your skin. If the implant gets inserted too deeply, removal can be tricky.
After the procedure
Your care provider will feel your arm to check the placement of the implant and have you do the same. An ultrasound or X-ray might be needed to check that the device got placed correctly.
A small bandage will cover the placement site. Also, a pressure bandage might be applied to lessen bruising. You can take off the pressure bandage in 24 hours. But keep the small bandage on and keep the area clean for 3 to 5 days.
It's common to have some bruising, pain, scarring or bleeding at the placement site.
Contact your care team if you notice:
- Breast lumps.
- Heavy or long-lasting vaginal bleeding.
- Signs or symptoms of a blood clot in your leg, such as constant pain and swelling in your calf.
- Signs or symptoms of jaundice, such as yellowing of the skin or whites of the eyes.
- Signs or symptoms of an infection at the placement site, such as tenderness, swelling, discharge or a change in skin color.
- Signs or symptoms of pregnancy at any time after the contraceptive implant is inserted.
Results
The contraceptive implant can prevent pregnancy for up to three years. It must be replaced at the three-year mark to continue protecting against unplanned pregnancy.
Your care team might suggest removing the contraceptive implant if you develop:
- Migraine with aura.
- Heart disease or stroke.
- Uncontrolled high blood pressure.
- Jaundice.
- Significant depression.
To remove the device, your provider will give you a shot of local anesthetic in your arm beneath the implant to numb the area. Next, a small cut will be made in the skin of your arm and the implant pushed to the surface. Once the tip of the implant can be seen, it will be grasped with forceps and taken out.
After the contraceptive implant is removed, the cut will be covered with a small bandage and pressure bandage. The removal procedure typically takes less than five minutes.
If you want, a new implant can be placed as soon as the original one is removed. Plan to use another type of birth control right away if you don't have a new contraceptive implant placed.