May 22, 2018
Andrew McKeon, M.D., M.B., B.Ch., and Sean J. Pittock, M.D., neurologists and members of the autoimmune neurology laboratory at Mayo Clinic in Rochester, Minnesota, answer questions about Mayo Clinic's new test.
Mayo Clinic has developed the first test available in the US to identify the myelin oligodendrocyte glycoprotein (MOG) antibody in patients' blood. How does this test help with the diagnosis of inflammatory demyelinating diseases?
The MOG antibody can distinguish a spectrum of autoimmune demyelinating diseases from multiple sclerosis (MS). Testing positive for the MOG antibody indicates that a patient doesn't have classical MS.
That's important because diseases associated with the MOG antibody — as well as diseases associated with the water channel aquaporin-4 (AQP4) antibody, which was discovered at Mayo Clinic — are commonly misdiagnosed as MS. Yet some MS medications have been reported to worsen diseases that mimic MS.
Mayo Medical Laboratories offers testing for both MOG and AQP4 antibodies, either separately or in combination. Combined testing allows for the most comprehensive evaluation for patients with recently diagnosed demyelinating diseases.
Which patients would benefit from this antibody testing?
Any patient who suddenly presents with vision loss, significant disk edema or recurrent optic neuritis should consider testing for both MOG and AQP4 antibodies. Patients who have more than one episode of optic neuritis absolutely should have testing. An optic neuritis seems to be a very common presentation of MOG.
Our test has great sensitivity and specificity. It uses a completely novel method of flow cytometry developed by Mayo Clinic to identify patient antibody binding to the MOG protein that is expressed on living cells.
MRI scans show enhancement of the left optic nerve and nerve sheath
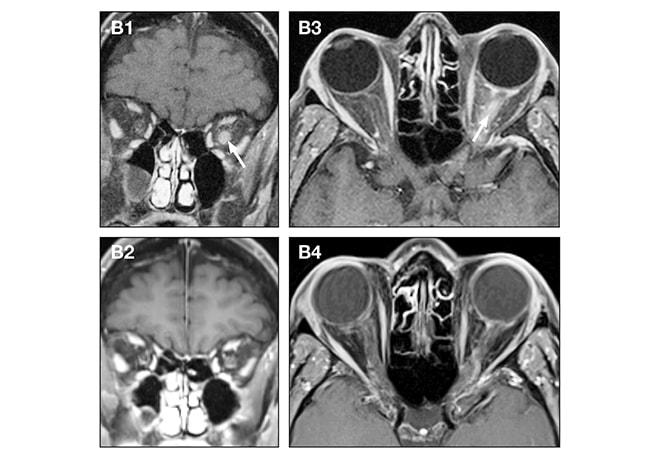
MRI scans show enhancement of the left optic nerve and nerve sheath
B1 and B3. Coronal and axial T1 post-contrast MRI scans show enhancement of the left optic nerve and nerve sheath (arrows) in an adult with recurrent optic neuritis. B2 and B4. Improvement of the left optic nerve lesions is visible four months later, after immunotherapy.
FLAIR image of adult patient demonstrates multifocal T2 hyperintense lesions
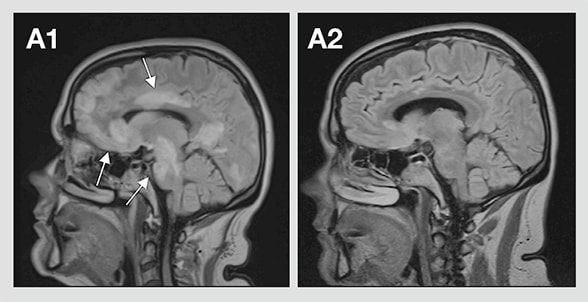
FLAIR image of adult patient demonstrates multifocal T2 hyperintense lesions
A1. Sagittal T2 fluid-attenuated inversion recovery (FLAIR) image of an adult patient demonstrates multifocal T2 hyperintense lesions in the frontal, occipital and temporal lobes, the corpus callosum, and the brainstem (arrows), consistent with acute disseminated encephalomyelitis. A2. Sagittal T2 FLAIR image nine months later after immunotherapy shows almost complete resolution of the T2 hyperintense lesions.
Which inflammatory disease phenotypes are associated with the MOG antibody?
Our work indicates that testing is positive in:
- 60 percent of pediatric patients and 40 percent of adult patients with acute disseminated encephalomyelitis
- 30 percent of patients with neuromyelitis optica who lack the AQP4 antibody
- 20 percent of patients with a longitudinally extensive transverse myelitis
- 10 percent of patients with recurrent optic neuritis and 2 percent of patients with a single optic neuritis
How does the presence of the MOG antibody affect disease course?
It may actually indicate a more favorable prognosis. For example, in patients with recurrent optic neuritis, the visual outcomes are better with MOG-associated disease than AQP4-associated disease. The diseases are bizarrely different — AQP4 targets astrocytes while MOG targets oligodendrocytes — yet the phenotypes look quite similar. We are planning a study to learn more about these phenotypes.
We've also found that the persistence of the MOG antibody is associated with disease relapses. Many of these patients will see a very rapid drop in MOG antibodies and test negative within six months. Those patients are at low risk of another attack. But if the antibody persists, the patient is at higher risk of another attack. This is a very interesting phenomenon and quite unusual in the field of autoimmune neurology.
What are the implications for treatment?
In general, we would treat each attack, maybe aggressively, with steroids. The persistence of the MOG antibody might indicate a need for medication to prevent disease relapse and possibly reduce disability progression. But whether all of these patients should be put on heavy-hitting immune suppression isn't 100 percent clear. Many of these patients might have one or two optic neuritis attacks over five years and have normal visual outcomes.
What other antibody tests might become available?
We expect to discover more antibodies. For example, we know that only about 20 percent of people with recurrent episodes of optic neuritis have the MOG or the AQP4 antibody. The other 80 percent of people behave very similarly clinically but don't have either of those biomarkers. This area is ripe for discovery.
How does the autoimmune neurology laboratory facilitate Mayo Clinic's translational science?
The lab is a multidisciplinary group that includes people with very strong clinical backgrounds as well as scientific backgrounds. Having that clinical expertise in a laboratory environment helps us focus on the needs of patients who will have this type of testing. The lab also has a large repository of specimens that allowed us to develop the MOG assay relatively quickly. This type of testing, for relatively common but treatable diseases, directly benefits patients.