Sept. 05, 2019
Imaging of dAVFs illustrates the missing-piece pattern
Imaging of dAVFs illustrates the missing-piece pattern
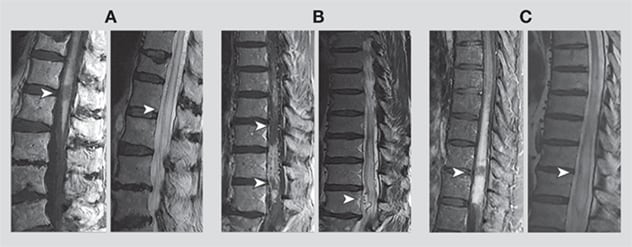
Imaging of three patients with spinal dural arteriovenous fistulas (dAVFs) illustrates the missing-piece pattern. A-C, left panels. Arrows indicate a T1-weighted gadolinium enhancement pattern with abrupt pieces of contrast enhancement missing. A-C, right panels. Arrows in the accompanying sagittal T2-weighted imaging show possible subtle flow voids in the first patient (A), prominent tortuous flow voids in the second patient (B) and no flow voids in the third patient (C), whose spinal dAVF was recognized only at the time of durotomy. Imaging reprinted with permission from JAMA Neurology.
Mayo Clinic has identified a unique radiographic finding that facilitates earlier diagnosis of spinal dural arteriovenous fistulas (dAVFs), paving the way to improved treatment outcomes. In the unusual MRI pattern, abrupt segments are seemingly missing amid an area of intense whole-cord contrast enhancement — a finding that Mayo Clinic calls the missing-piece sign.
"When neurologists see enhancement on spinal MRI, their automatic first thoughts are inflammation or tumor. But enhancement can be due to many other etiologies, including a fistula," says Nicholas (Nick) L. Zalewski, M.D., a neurologist at Mayo Clinic in Rochester, Minnesota.
Spinal dAVFs are rare, with heterogenous clinical and radiologic features that can lead to misdiagnoses of inflammatory or neoplastic myelopathies. Although clinical deficits resulting from spinal dAVFs can be reversible, a delay in diagnosis is associated with severe permanent morbidity.
"Early detection and treatment are key to achieving optimal outcomes," says Jonathan L. Carter, M.D., a neurologist at Mayo Clinic in Phoenix/Scottsdale, Arizona. "Mayo Clinic has a great deal of experience managing a variety of spinal cord disorders, which helps us to differentiate and arrive at an accurate diagnosis."
Spinal cord angiogram, a tool critical for precisely diagnosing and locating a spinal dAVF, is performed by a designated group of neuroradiologists and neurosurgeons. "Finding the fistula is often the most difficult part. Once a spinal dAVF is correctly diagnosed, we have the ability to treat it with embolization or surgery, as needed," says Giuseppe Lanzino, M.D., a neurosurgeon at Mayo Clinic's campus in Minnesota. "With timely intervention, patients can have a remarkable recovery."
Finding the missing piece
Although rare, spinal dAVFs are underdiagnosed. The initial clinical signs and symptoms can include:
- Gradually progressive myelopathy, particularly in an older patient
- Myelopathy features affecting the lower thoracic region into the conus
- Deficits that suddenly worsen with exertion or Valsalva
Tortuous flow voids — another MRI finding associated with spinal dAVF — are classic but not universally present.
"It is very important to consider spinal dAVF in the differential diagnosis when evaluating patients with progressive myelopathy and abnormalities in spinal cord MRI because misdiagnosis can lead to morbidity and to unnecessary diagnostic testing and treatment," says Iris (Vanessa) V. Marin Collazo, M.D., a neurologist at Mayo Clinic in Jacksonville, Florida. "A high index of suspicion, excellent diagnostic tools and criteria, and an experienced, multidisciplinary team are essential."
Mayo Clinic's extensive experience with spinal cord diseases facilitated the discovery of the spinal dAVF radiologic clue. That discovery — reported in the Dec. 1, 2018, issue of JAMA Neurology — arose from an earlier Mayo Clinic study that proposed diagnostic criteria for spinal cord infarction. For that large case series, published in the Jan. 1, 2019, issue of JAMA Neurology, Mayo researchers reviewed cases associated with other causes of myelopathy, including spinal dAVF, as a control.
"When looking over case after case of spinal dAVF, this recurrent imaging pattern stuck out as bizarre," Dr. Zalewski says. "In these cases, we noticed that the entire circumference of the spinal cord showed enhancement for at least a couple of segments, but within that, there was a very abrupt missing segment of enhancement. It's almost as if someone took a chunk out of the spinal cord in one very focal spot. We had never quite seen this before."
The researchers suspect the peculiar imaging pattern is due to inconsistency of the intrinsic venous system of the spinal cord, with the abrupt segments without enhancement potentially having better venous egress routes than the adjacent cord.
The missing-piece sign can potentially result in earlier angiography to confirm a diagnosis of spinal dAVF. Mayo Clinic often initially uses MR angiogram (MRA), followed by angiogram. "MRA of the spinal canal can usually at least tell us the probable location of the fistula," Dr. Zalewski says. "Our interventional radiologist can then perform an angiogram at more targeted levels to find the exact location and consider embolization, rather than going to every single level in the spinal canal, which can take some time."
Spinal angiogram is a highly specialized procedure. "Locating a malformation in these tiny blood vessels requires experience," Dr. Lanzino says. "At Mayo Clinic, we have seen quite a few patients who had an angiogram elsewhere, but the fistula wasn't detected."
In Mayo Clinic's missing-piece study, five of 19 patients with the imaging sign required more than one angiogram to confirm a diagnosis of spinal dAVF. "If there is a strong suspicion clinically and radiologically for spinal dAVF — even if the fistula isn't obvious on a first screen — it's important to be vigilant and look harder," Dr. Zalewski says.
The consequences of delayed diagnosis of spinal dAVF were demonstrated by Mayo Clinic researchers in a study published in the February 2016 issue of the American Journal of Neuroradiology. The researchers reviewed initially misdiagnosed cases of spinal dAVF ultimately seen at Mayo Clinic between 2000 and 2014. The median delay before an angiographically confirmed diagnosis of spinal dAVF was six months.
All 53 patients in the cohort could ambulate independently at the time of their initial imaging studies demonstrating a spinal dAVF. More than half of the patients in the cohort required a wheelchair or a walker by the time the spinal dAVF was recognized. This progression of disability was usually irreversible; only one-third of patients who required a wheelchair at the time of spinal dAVF diagnosis were able to ambulate with a walker 90 days after treatment.
Finding specific answers
As a tertiary center, Mayo Clinic has experience with spinal epidural AVFs, a rare but treatable subtype of spinal dAVF. Spinal epidural AVFs are the result of a fistulous connection between the branches of the paraspinal or paravertebral arterial system and the epidural venous plexus. Like spinal dAVFs, spinal epidural AVFs often initially present with vague symptoms, such as leg dysesthesia and exertional leg weakness, that progress slowly to severe myelopathy with paraplegia and sphincter dysfunction.
"A multidisciplinary approach and an experienced team are critical, as a neurosurgeon might see only a handful of these cases over the course of a career," Dr. Lanzino says. As reported in the May 2017 issue of the Journal of Neurosurgery: Spine, Mayo Clinic successfully treated the majority of spinal epidural AVFs seen there between 2005 and 2015 with endovascular therapy alone. Among all the patients treated for spinal epidural AVF, nearly two-thirds experienced some improvement in disability and motor function after treatment, and more than half of treated patients had some improvement in sensory symptoms.
"At Mayo Clinic, we try our absolute hardest to give a definitive diagnosis for patients," Dr. Zalewski says. "We look for additional clues in tough cases, and we are able to confer and collaborate with our colleagues. Rather than putting patients into a vague category, our goal is always to find a very specific answer."
For more information
Zalewski NL, et al. Unique gadolinium enhancement pattern in spinal dural arteriovenous fistulas. JAMA Neurology. 2018;75:1542.
Zalewski NL, et al. Characteristics of spontaneous spinal cord infarction and proposed diagnostic criteria. JAMA Neurology. 2019;76:56.
Brinjikji W, et al. Clinical outcomes of patients with delayed diagnosis of spinal dural arteriovenous fistulas. American Journal of Neuroradiology. 2016;37:380.
Nasr DM, et al. Clinical presentation and treatment outcomes of spinal epidural arteriovenous fistulas. Journal of Neurosurgery: Spine. 2017;26:613.