Aug. 20, 2021
Mayo Clinic uses biomarker testing to evaluate for brain changes related to Alzheimer's disease in patients with diagnoses of mild cognitive impairment or dementia. The biomarker test results, which are generally available within a week, can increase diagnostic accuracy above the 70% to 80% level achieved through standard clinical and cognitive assessments for Alzheimer's disease.
"While Alzheimer's disease is a difficult diagnosis, having that information helps patients and their families plan for the future. It also allows patients to avoid unnecessary testing for other, less common conditions," says Gregory (Gregg) S. Day, M.D., a behavioral neurologist at Mayo Clinic in Jacksonville, Florida.
Mayo Clinic's biomarker testing, which uses cerebral spinal fluid (CSF), is intended for patients who have already been evaluated for Alzheimer's disease using standard clinical tools and approaches. "The biomarker test should not be used to screen for memory problems or Alzheimer's disease in a person without symptoms," Dr. Day says.
Negative for beta- amyloid

Negative for beta- amyloid
Pittsburgh compound-B positron emission tomography (PiB-PET) indicates no beta-amyloid buildup.
Positive for amyloid
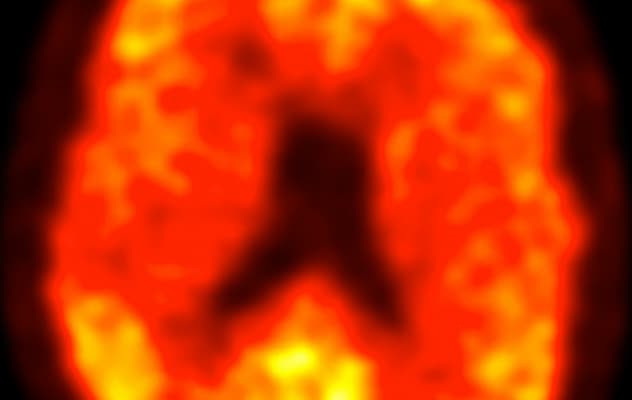
Positive for amyloid
Increased red and yellow areas in this PiB-PET scan represent beta-amyloid buildup.
The test includes CSF assays for beta-amyloid, total tau and phosphorylated tau. Although beta-amyloid can be detected with positron emission tomography (PET), that testing is costly.
"Together our three spinal fluid biomarkers can predict PET scan findings," Dr. Day says. "The biomarker test results can help us match patients to treatments for the symptoms of Alzheimer's disease, although those treatments are very limited."
Mayo Clinic's biomarker testing uses strict protocols to ensure accurate results. CSF must be drawn directly into a special tube required by the highly sensitive machine that processes the test. "Transferring spinal fluid across tubes leaves some of the important proteins behind and changes the results," Dr. Day says. "Avoiding unnecessary manipulation of the fluid is one reason we are able to obtain reliable, reproducible results."
Dr. Day notes that most patients want to know as much as possible about their diagnoses of Alzheimer's disease. "That information may be especially helpful for individuals in their 60s or younger. It might affect decisions about retirement or travel," he says. "An early diagnosis of Alzheimer's disease might also allow patients to participate in research or clinical trials that are open only to individuals in the very mild or mild stages of the disease."
Patient-centered research
Mayo Clinic is committed to finding biomarkers for neurological diseases. In partnership with basic scientists, clinicians are searching for additional biomarkers that further the understanding of Alzheimer's disease and how it affects individual patients.
"After receiving a diagnosis of Alzheimer's disease, every patient asks us two questions: 'How severe is it, and what does this mean for me in one or two years?' " Dr. Day says. "We can't yet answer those questions with the biomarkers that we have."
Mayo Clinic is also working to develop biomarkers for conditions that can mimic or coexist with Alzheimer's disease, such as Lewy body dementia and vascular disease. Another focus is identifying biomarkers in blood that can provide information now detectable only in CSF.
Dr. Day notes the usefulness of biomarkers for enrolling patients in studies that might eventually change how and when people are treated. Mayo Clinic is participating in the large, multicenter AHEAD 3-45 study that is considering the possibility of treating Alzheimer's disease before symptoms emerge — thereby preventing the development of mild cognitive impairment and dementia in at-risk individuals.
"That kind of study would have been unthinkable 10 or 15 years ago," Dr. Day says. "We're able to do it now only because we can confidently use biomarkers to identify Alzheimer's disease during life."
For more information
Eisai Inc. AHEAD 3-45 Study: A Study to Evaluate Efficacy and Safety of Treatment With Lecanemab in Participants With Preclinical Alzheimer's Disease and Elevated Amyloid and Also in Participants With Early Preclinical Alzheimer's Disease and Intermediate Amyloid. ClinicalTrials.gov.