Jan. 17, 2020
Endocrinologists play an important role in prescribing and managing hormone therapy for gender dysphoria, and guidelines regarding the care of transgender patients are available. There is now a growing recognition that long-term hormone therapy may impact the risk of a number of chronic diseases, including osteoporosis.
Estrogen is the dominant sex steroid regulating bone resorption and formation in both cisgender (identify with sex assigned at birth) males and cisgender females. "However, evolving evidence has highlighted a potential independent role for androgens in regulating cancellous bone remodeling (as in the vertebrae) in cisgender males, with estrogen likely mainly regulating cortical remodeling (as in the long bones)," clarifies Sundeep Khosla, M.D., Endocrinology, Diabetes, Metabolism and Nutrition, at Mayo Clinic in Rochester, Minnesota.
Working models for sex steroid action
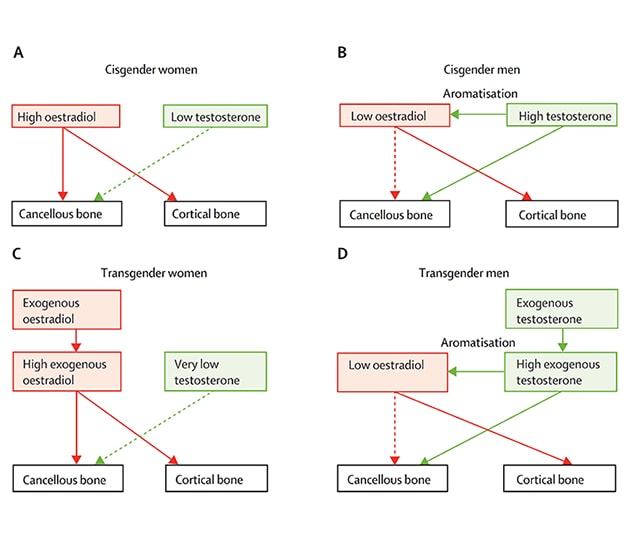
Working models for sex steroid action
Working model for sex steroid action on cancellous versus cortical bone in (A) cisgender females and (B) cisgender males. Panels (C) and (D) show the analogous models in transgender females and males, respectively, placing the models in the context of exogenous hormone therapies. Image reprinted with permission from The Lancet Diabetes & Endocrinology.
Caroline J. Davidge-Pitts, M.B., B.Ch., Endocrinology, Diabetes, Metabolism and Nutrition, at Mayo Clinic in Rochester, Minnesota, explains: "In cisgender females prior to menopause, the high endogenous ovarian estrogen levels inhibit excessive bone resorption and help maintain bone formation in both cancellous and cortical bone. The role of the low androgen levels in regulating bone remodeling in cisgender females remains unclear, although evidence from androgen receptor knockout mice indicates that androgens may have some effects on cancellous, but not cortical, bone remodeling.
"By contrast, in cisgender males, the relatively low estradiol levels coming both from the testes and via peripheral aromatization of testosterone are likely insufficient to fully prevent excessive bone remodeling and bone loss in cancellous bone, perhaps because cancellous bone also contains considerable amounts of estrogen receptor (ER) β, which serves to antagonize ERα action and thus make cancellous bone relatively resistant to estrogen. Cortical bone, which expresses little or no ERβ, may be much more sensitive to estrogen, which appears to mainly target cortical bone in males. However, the existing mouse and human data are consistent with a significant effect of testosterone at the levels found in cisgender males, in the absence of aromatization to estrogen, in preventing loss of cancellous bone. In addition to having this effect, testosterone likely plays an important role in driving periosteal bone formation and the larger bone size in cisgender males achieved during puberty.
"In this context, one can then make some predictions regarding the skeletal effects of hormone therapy and compare these to the existing clinical data, with the caveat that most studies to date have utilized dual energy X-ray absorptiometry (DXA), which cannot distinguish cancellous bone from cortical bone.
"Thus, in transgender women (sex assigned male at birth, identify as female), the high exogenous estradiol levels should be sufficient to protect both cancellous and cortical bone, although it is possible that there may be some deficits in cancellous bone if testosterone levels are very low due to gonadal suppression or orchiectomy. In transgender men (sex assigned female at birth, identify as male), there should be sufficient exogenous testosterone to protect cancellous bone as well as aromatization of testosterone to estradiol to prevent cortical bone loss. These effects of sex steroids are likely amplified during puberty, as sex steroids clearly regulate bone mass acquisition during this period. Importantly, fusion of the growth plate is dependent on estrogen in both females and males."
Dr. Khosla highlights: "This working model would predict that, assuming compliance with hormone therapy, the skeleton should be relatively well protected in both transgender women and transgender men."
Compared with cisgender men, transgender women have lower bone mass and cortical size even prior to initiation of hormone therapy, suggesting sex steroid-independent effects in these individuals. "These individuals are more likely to have vitamin D deficiency and less likely to be involved in sport than cisgender men," says Dr. Davidge-Pitts.
Although bone mineral density (BMD) is generally preserved in both transgender women and transgender men, there are sparse data on fracture risk. In the largest fracture study to date, which included 2,023 transgender women and 1,036 transgender men, fracture risk was not increased in transgender men when compared with either reference cisgender men or cisgender women, but it tended to be increased in transgender women younger than 50 years when compared with age-matched reference cisgender women but not when compared with age-matched reference cisgender men.
In transgender women older than 50 years, fracture risk was similar to that of age-matched reference cisgender women, but was increased almost twofold compared with that of age-matched reference cisgender men. "Whether this risk differential is related to the underlying skeletal deficits in transgender women even prior to starting hormone treatment noted earlier remains to be determined," says Dr. Khosla. "It should be noted, however, that the observation period for this fracture cohort was only three years, so longer term fracture incidence data in transgender individuals are clearly needed. The bone-specific long-term effects of puberty blockers and hormone therapy initiated in adolescence are yet to be fully determined."
Dr. Khosla stresses that the 2019 International Society for Clinical Densitometry guidelines have addressed the use of BMD determined with DXA in transgender and gender-nonconforming individuals. Baseline BMD measurement recommendations are similar to those in cisgender individuals. Follow-up BMD measurement can be considered in individuals with baseline low bone density, suboptimal dosing of or non-compliance with hormone therapy, or plans to discontinue hormone therapy; in those who are at increased risk of fracture; and in the setting of agents that suppress puberty.
It is also recommended that T-scores be calculated using the uniform Caucasian female normative database. Z-scores should be calculated using the affirmed gender normative database; however, providers could also request the normative database for sex assigned at birth. Additionally, nonbinary individuals should have Z-scores calculated using the normative database for sex assigned at birth.
Dr. Davidge-Pitts concludes: "Regarding bone health in transgender and gender-nonconforming individuals, there remain several important research and clinical issues that are unresolved. Further studies are needed to address these gaps."
The most current evidence was reviewed by Dr. Davidge-Pitts, Dr. Khosla and a research team at Mayo Clinic in The Lancet Diabetes & Endocrinology and Maturitas in 2019.
For more information
Khosla S, et al. Skeletal considerations in the medical treatment of transgender people. The Lancet Diabetes & Endocrinology. 2019;7:893.
Davidge-Pitts C, et al. Transgender bone health. Maturitas. 2019;127:35.
The International Society for Clinical Densitometry.