Oct. 18, 2018
Management of large colonic polyps via endoscopic procedures is a rapidly changing field. Once surgically managed, large (greater than 2 cm) colorectal lesions can now be safely and effectively treated endoscopically, most commonly with endoscopic mucosal resection (EMR). When compared with traditional surgical excision, EMR has several associated benefits, including high efficacy and patient satisfaction rates, lower costs and complication rates, rapid recovery, and the ability to preserve normal gut functions.
According to therapeutic endoscopists at Mayo Clinic, recent technical advances have broadened the scope of what can be accomplished using EMR.
"Experienced operators can use EMR to remove large polyps, even near-circumferential lesions, which traditionally have required surgery. A number of factors come into play when assessing whether a lesion is suitable for EMR, including lesion location and access, mucosal features and adequate submucosal fluid lift," says Louis M. Wong Kee Song, M.D., a therapeutic endoscopist at Mayo Clinic's campus in Rochester, Minnesota. "EMR can also be considered for lesions that involve to some extent the appendiceal orifice or ileocecal valve, but these require specialized techniques and expertise."
EMR for colorectal polyps
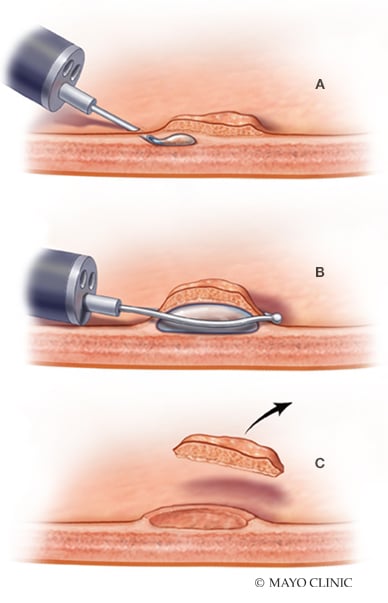
EMR for colorectal polyps
Endoscopic mucosal resection (EMR) for colorectal polyps. A. Submucosal fluid injection. B. Snare resection. C. Post-resection defect.
To facilitate the removal of these larger lesions, endoscopists typically inject fluid into the submucosal space. This technique generates a safe fluid cushion between the mucosal and the muscular layers, which is designed to reduce thermal injury and the risk of perforation, and to facilitate en bloc resection or resection in fewer pieces.
The most commonly used fluid for this procedure is a 0.9 percent sodium chloride (NaCl) solution stained with a dye, such as methylene blue or indigo carmine. One of the disadvantages associated with this solution is its rapid reabsorption rate, often necessitating multiple injections and extending the length of time required to perform the procedure.
Recognizing these challenges, Mayo Clinic researchers recently participated in a multicenter randomized double-blind trial comparing the efficacy and safety of a new Food and Drug Administration-approved solution for submucosal injection (SIC-8000) with standard saline solution in patients undergoing EMR of large colorectal lesions.
SIC-8000 is a low-viscosity solution developed to provide a long-lasting cushion to facilitate endoscopic resection maneuvers. A pre-mixed, ready-to-use blend of a synthetic copolymer and methylene blue, the solution is designed to delay fluid from leaving the injected area.
"Our study yielded both technically and clinically relevant results," explains Michael B. Wallace, M.D., a therapeutic endoscopist at Mayo Clinic's campus in Florida.
Study methods
A total of 226 patients undergoing EMR at five tertiary medical centers for nonpedunculated colorectal lesions ≥ 20 mm were enrolled in the study and randomized between the two submucosal agents. Endoscopists and patients were blinded to the type of submucosal solution used.
Primary endpoints included total volume of solution required to complete the procedure; the volume per lesion size; and the length of time required to complete the resection, measured from the first injection to the last piece of lesion excised. Secondary endpoints included Sydney Resection Quotient (SRQ), multiple other EMR outcomes and the rate of adverse events (AEs).
Study results
Results, published in Gastrointestinal Endoscopy in 2018, included data from 211 patients in which EMR was completed, comparing the two groups' study endpoints and data related to AEs obtained from a telephone follow-up performed 30 days post-procedure.
Efficacy
- Total volume needed for EMR was significantly less in the SIC-8000 group (16.1 ± 9.8 mL) than in the saline solution group (31.6 ± 32.0 mL), P < 0.001.
- Average volume per lesion size was 0.5 ± 0.3 mL/mm in the SIC-8000 group and 0.9 ± 0.6 mL/mm in the saline solution group, P < 0.001.
- The mean time to completely resect lesions was lower for the SIC-8000 group (19.1 ± 16.8 minutes) compared with the saline solution group (29.7 ± 68.9 minutes), P = 0.1.
EMR outcomes
- The SRQ was significantly higher in the SIC-8000 group (10.3 ± 8.1) compared with the saline solution group (8.0 ± 5.7), P = 0.04
- The number of resected pieces was lower in the SIC-8000 group (5.7 ± 6.0) compared with the saline solution group (6.5 ± 5.04), P = 0.052.
Safety profile
- The rate of AEs was similar between the two groups (SIC-8000, 18.6 percent; saline solution, 17 percent), and none of the serious AEs (SIC-8000 group, 8.8 percent; saline solution group, 10.7 percent) were related to the study treatment.
Study limitations
- The predictive limits of using SRQ as a proxy for a more favorable EMR clinical outcome.
- The fact that procedures were performed in tertiary care center settings by highly experienced endoscopists, which may make the results difficult to replicate in other settings.
"Overall, the new solution outperformed saline solution in outcomes related to efficacy and multiple other clinically relevant measures, such as the time required to perform the procedure," says Dr. Wallace. "And it equaled the performance of saline solution for safety-related outcomes. The technical and clinical superiority of SIC-8000 demonstrated in this clinical trial is also consistent with results obtained in previous animal studies."
Endoscopic removal of large lesions remains a technically challenging procedure that requires considerable skill and experience. Drs. Wallace and Wong Kee Song caution that it's important to be aware of factors that prevent proper lifting of polyps, including injection of tattoo material underneath or too close to polyps, and any use of snare or extensive biopsy prior to referral for EMR. However, new techniques and tools that help reduce the complexity and length of time associated with these procedures are a welcome development. These advances mean that in the hands of skilled endoscopists, nearly all noninvasive polyps (of any size) can be safely and effectively removed with endoscopic resection and thus do not require surgical referral.
For more information
Repici A, et al. A novel submucosal injection solution for endoscopic resection of large colorectal lesions: A randomized, double-blind trial.Gastrointestinal Endoscopy. 2018;88:527.