Nov. 19, 2021
Over the last decade, mortality from kidney cancer has remained unchanged despite advancements in early detection and treatment for advanced disease. Mayo Clinic researchers previously developed a risk stratification model to predict cancer-specific survival rates, which can range widely based on various features. However, the biological mechanism for heterogeneity in cancer-specific outcomes is still the subject of considerable investigation.
A study published in Urologic Oncology: Seminars and Original Investigations in 2021 reports that a newly investigated tumor tissue expression biomarker is Bcl-2 interacting mediator of cell death (BIM), a downstream pro-apoptotic signaling molecule in tumor reactive CD8+ T cells that is upregulated with the interaction of PD-L1 and PD-1.
BIM plays a crucial role in deletion of self-reactive immune cells. PD-L1 deficiency has been shown to significantly reduce BIM levels in CD8+ T cells and decrease T cell apoptosis. This finding suggests that BIM has an important role in PD-L1-induced T cell apoptosis.
Given the recent success of checkpoint inhibitor therapy in renal cell carcinoma, the use of BIM as a biomarker is promising, although there have been limited studies of this molecule for renal cell carcinoma.
"The identification of primary tumor tissue biomarkers such as BIM could improve prediction of cancer-specific survival in patients with metastatic clear cell renal carcinoma and improve on our previously reported clinical metastases score," notes Elizabeth N. Bearrick, M.D., a Urology resident at Mayo Clinic in Rochester, Minnesota.
Methodology
The authors of the study assessed tissue blocks from patients who underwent nephrectomy and developed metastatic disease. These tissue blocks were assessed for expression of various biomarkers, including BIM. Biomarkers that were significantly associated with cancer-specific survival after adjusting for the metastases score were used to develop a biomarker-specific model.
Results
The institutional database included 602 patients who received nephrectomy between 1990 and 2004, including:
- 192 (32%) with metastases at diagnosis
- 410 (68%) who developed metastases after nephrectomy
Median follow-up was 9.6 years (IQR 4.2-12.8), during which 504 patients died of renal cell carcinoma.
Low BIM and low survivin
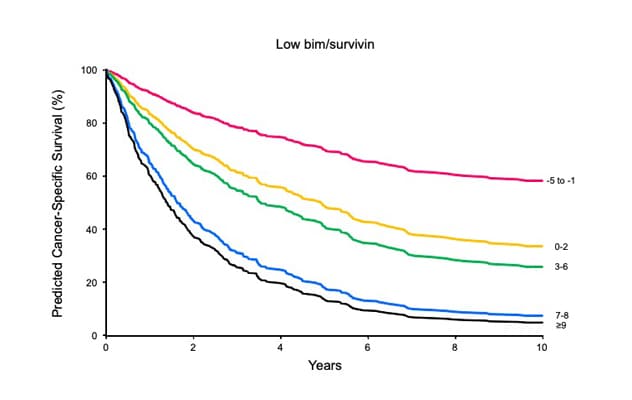
Low BIM and low survivin
Patients with high BIM and high survivin expression had significantly worse cancer-specific survival compared with patients with low BIM and low survivin expression within specific metastases score ranges.
High BIM and high survivin
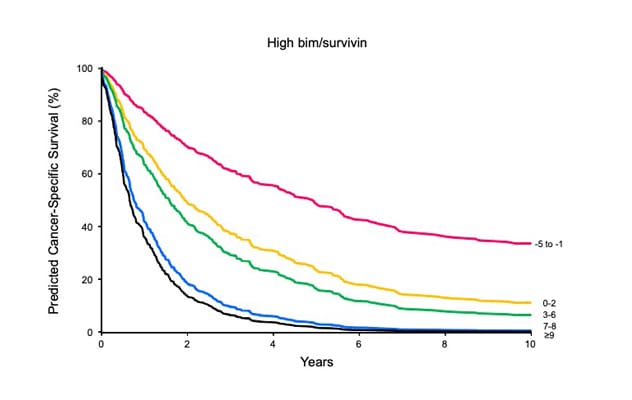
High BIM and high survivin
The associations of combined BIM and survivin expression with death from renal cell carcinoma after adjusting for the metastases score suggest that patients whose primary tumors contained high levels of both BIM and survivin experienced the worst outcomes following distant metastases.
Expression of several biomarkers studied, including BIM, was significantly associated with cancer-specific survival after adjusting for the metastases score, and they were eligible for biomarker-specific model inclusion. After variable selection, high BIM, high survivin and the metastases score were retained in the final multivariable model.
Conclusions
Ultimately the researchers were able to create a prognostic model combining the previously developed clinical metastases score and two primary tumor tissue expression biomarkers, BIM and survivin, for patients with metastatic renal cell carcinoma who underwent nephrectomy.
"We find these results to be extremely promising," says Bradley C. Leibovich, M.D., Urology, Mayo Clinic in Minnesota, and senior author of the study. There has historically been significant heterogeneity in cancer-specific outcomes. Our hope is that this model helps tailor risk-stratified management, with earlier intervention and consideration for novel therapeutic agents and clinical trials, for high-risk patients."
For more information
Bearrick EN, et al. Creation of a primary tumor tissue expression biomarker-augmented prognostic model for patients with metastatic renal cell carcinoma. Urologic Oncology: Seminars and Original Investigations. 2021;39:135.