Overview
Copper IUD (ParaGard)

Copper IUD (ParaGard)
The copper IUD (ParaGard) contains no hormones and prevents pregnancy for up to 10 years.
ParaGard is a type of long-term birth control called an intrauterine device, also known as an IUD. ParaGard releases copper into the uterus to prevent pregnancy. It doesn't release hormones like other IUDs do.
ParaGard is a T-shaped plastic frame with copper wire coiled around it. A healthcare professional places the device into the uterus. The copper affects sperm function and movement. This helps keep sperm from reaching the uterus and joining with an egg, also called fertilization.
ParaGard is the only copper IUD available in the United States. It can prevent pregnancy for up to 10 years.
Why it's done
ParaGard is a safe, effective and long-term way to prevent pregnancy. Adults who haven't gone through menopause can use it, as can teenagers.
Among its upsides, ParaGard:
- Gets rid of the need to interrupt sex to use a condom or other birth control methods.
- Can stay in place for up to 10 years.
- Can be removed at any time.
- Can be used while breastfeeding.
- Doesn't carry the risk of side effects related to hormonal birth control, such as blood clots.
- Can be used for emergency birth control if a healthcare professional inserts it within five days of having sex without protection.
ParaGard isn't right for everyone. Your healthcare professional likely will recommend a different type of birth control if you:
- Are already pregnant or might be pregnant, because ParaGard can raise the risk of a miscarriage.
- Have conditions of the uterus that make it hard to insert ParaGard and keep it in place. These conditions include large growths called fibroids that aren't cancer.
- Have a pelvic infection such as pelvic inflammatory disease or endometriosis.
- Are waiting to get treatment for uterine or cervical cancer.
- Have vaginal bleeding without a clear cause.
- Are allergic to any part of the ParaGard device, such as the copper wire.
- Have a condition called Wilson's disease that causes too much copper to build up in the liver, brain and other vital organs.
Risks
Less than 1% of people who use ParaGard get pregnant in the first year of typical use. Over time, the risk of pregnancy in people who use ParaGard stays low.
If you do become pregnant while using ParaGard, you're at high risk of an ectopic pregnancy. This is when a fertilized egg attaches and grows outside the uterus, usually in a fallopian tube. The fertilized egg can't survive. And the growing tissue may cause life-threatening bleeding without treatment. But ParaGard prevents most pregnancies. So the risk of having an ectopic pregnancy is lower than it is for sexually active people who don't use birth control.
ParaGard doesn't protect you from sexually transmitted infections, also called STIs. If you think you're at risk of getting an STI, use a condom each time you have sex.
Side effects of ParaGard can include:
- Irregular bleeding and spotting between periods.
- Cramps.
- Serious menstrual pain and heavy bleeding, especially during the first few months of use.
It's also possible for part or all of the ParaGard device to fall out of the uterus by itself. You might not feel this if it happens.
ParaGard may be more likely to fall out if you:
- Have heavy or long-lasting periods.
- Have serious menstrual pain.
- Have had an IUD that fell out by itself in the past.
- Are younger than age 20.
- Had the IUD placed right after childbirth or after an abortion later in pregnancy.
How you prepare
A healthcare professional can insert ParaGard anytime during a typical menstrual cycle. If you just had a baby, your healthcare professional may recommend waiting about six weeks after delivery before inserting ParaGard.
Before inserting ParaGard, your healthcare professional reviews your overall health and does a pelvic exam. You may have a pregnancy test to confirm you're not pregnant. And you may be screened for STIs.
Taking a nonsteroidal anti-inflammatory drug, also called an NSAID, 1 to 2 hours before ParaGard is inserted can help ease cramping.
What you can expect
ParaGard placement
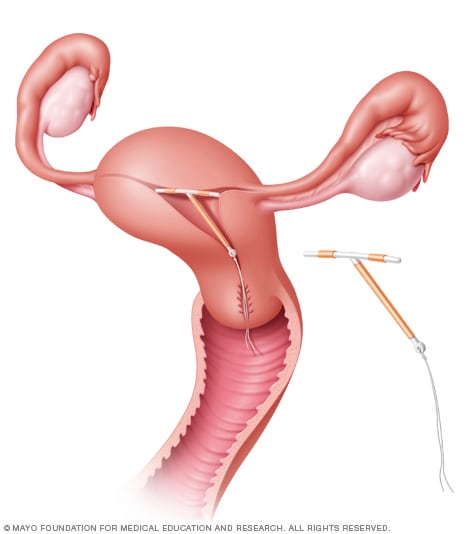
ParaGard placement
The ParaGard IUD can remain in place in the uterus for up to 10 years. Short strings connected to the device extend beyond the cervix into the vagina. The strings help a healthcare professional remove the device.
Most often, you go to a healthcare professional's office to get ParaGard inserted.
During the procedure
Your healthcare professional uses a plastic or metal-hinged tool called a speculum to spread open the walls of your vagina. The vagina and the canal that connects the vagina to your uterus, called the cervix, are cleaned with an antiseptic solution. A special instrument may be used to gently align your cervix and uterine cavity. Next, your healthcare professional folds down ParaGard's arms and places the device inside a tube.
The tube is inserted into your cervix, and ParaGard is carefully placed in your uterus. When the insertion tube is removed, ParaGard stays in place. Your healthcare professional trims ParaGard's attached strings so that they don't protrude too far into the vagina. The length of the strings may be noted as well. The strings help your healthcare professional remove ParaGard when you want the IUD taken out.
When ParaGard is placed in the uterus, it's possible to have side effects such as:
- Dizziness.
- Upset stomach.
- Fainting.
- Slowed heart rate.
Your healthcare professional likely will suggest that you stay lying down for a few minutes to let these side effects pass.
Some people have seizures when ParaGard is inserted. And rarely, the IUD punctures the uterine wall or cervix. Talk with your healthcare professional about all the risks before you get ParaGard inserted. Ask how health concerns such as punctures are managed.
After the procedure
About a month after ParaGard is inserted, you may get a follow-up healthcare checkup. Your healthcare professional checks to make sure the device hasn't moved.
While you're using ParaGard, call your healthcare professional right away if you have:
- Pregnancy symptoms such as a missed period.
- Heavy vaginal bleeding.
- Vaginal discharge that smells bad.
- Pelvic pain that gets worse.
- Serious pain or tenderness in the stomach area.
- Fever without a clear cause.
- Possible exposure to an STI.
It's also important to call your healthcare professional right away if you think your ParaGard is no longer in place. Call your healthcare professional if:
- You have irregular bleeding or bleeding after sex.
- Sex is painful for you or your partner.
- The ParaGard strings are missing or suddenly seem longer.
- You feel part of the device at your cervix or in your vagina.
Your healthcare professional can check the location of ParaGard and remove it if needed.
Removal
Most often, you go to your healthcare professional's office to have ParaGard removed during a pelvic exam. Your healthcare professional likely will use a tool called forceps to grasp the device's strings and gently pull. The device's arms fold upward as it's removed from the uterus.
Light bleeding and cramping are common during removal. It's also possible to have the same side effects that are linked to having the device inserted.
Feb. 15, 2025