Overview
Deep brain stimulation (DBS) involves implanting electrodes within areas of the brain. The electrodes produce electrical impulses that affect brain activity to treat medical conditions. The electrical impulses also can affect cells and chemicals within the brain that cause medical conditions.
A pacemakerlike device controls the amount of electrical activity in deep brain stimulation. The device is placed under the skin in the upper chest. A wire that travels under the skin connects this device to the electrodes in the brain.
Deep brain stimulation is commonly used to treat a number of conditions, such as:
- Parkinson's disease.
- Essential tremor.
- Conditions that cause the muscles to contract, known as dystonia, such as Meige syndrome.
- Epilepsy.
- Tourette syndrome, which causes repetitive movements and sounds.
- Obsessive-compulsive disorder.
Deep brain stimulation also is being studied as a potential treatment for:
- Quick movements that look like exaggerated fidgeting, known as chorea, such as in Huntington's disease.
- Long-lasting pain.
- Cluster headache.
- Dementia.
- Depression.
- Addiction.
- Obesity.
Products & Services
Why it's done
Deep brain stimulation is an established treatment for people with movement disorders. These conditions include essential tremor, Parkinson's disease and dystonia. DBS also is used for psychiatric conditions such as obsessive-compulsive disorder and Tourette syndrome. And deep brain stimulation has been approved by the U.S. Food and Drug Administration to reduce seizures in hard-to-treat epilepsy.
Deep brain stimulation is used in people whose symptoms aren't controlled with medicines.
Risks
Deep brain stimulation is generally considered to be low risk. But any type of surgery can potentially cause complications. Also, using electrical impulses in the brain can cause side effects.
Surgery risks
Deep brain stimulation involves making small holes in the skull to implant the electrodes into brain tissue. The procedure also includes surgery to implant a pacemaker-like device under the skin in the chest. This device contains the batteries needed for deep brain stimulation. Complications of surgery may include:
- Misplacement of the wires with the electrodes, known as leads.
- Bleeding in the brain.
- Stroke.
- Infection.
- Trouble breathing.
- Nausea.
- Heart problems.
- Seizures.
Possible side effects after surgery
Side effects of deep brain stimulation may include:
- Seizures.
- Infection.
- Headache.
- Confusion and trouble concentrating.
- Stroke.
- Hardware complications, such as an eroded lead wire.
- Short-term pain and swelling at the implantation site.
A few weeks after the surgery, your health professional turns on the device. Then, you and your health professional work on finding the settings that work best for you. Some settings may cause side effects, but these often improve with further changes to the settings of your device.
There have been rare reports that deep brain stimulation affects the movements needed for swimming. Talk to a healthcare professional before planning to swim and make sure to take water safety precautions.
Possible side effects of deep brain stimulation
After the device is turned on, you might experience these side effects:
- Numbness or tingling.
- Muscle tightness of the face or arm.
- Trouble with speech.
- Trouble with balance.
- Lightheadedness.
- Vision changes, such as double vision.
- Mood changes, such as anger, mania and depression.
How you prepare
Deep brain stimulation is a serious procedure. If you're eligible for this therapy, weigh the risks and potential benefits with your healthcare team.
Before surgery, you'll likely need medical tests to make sure that deep brain stimulation is a safe and good option for you. You also may need brain-imaging studies, such as an MRI, before the surgery. These studies help map the areas of your brain where the electrodes are implanted.
What you can expect
During the procedure
Deep brain stimulation
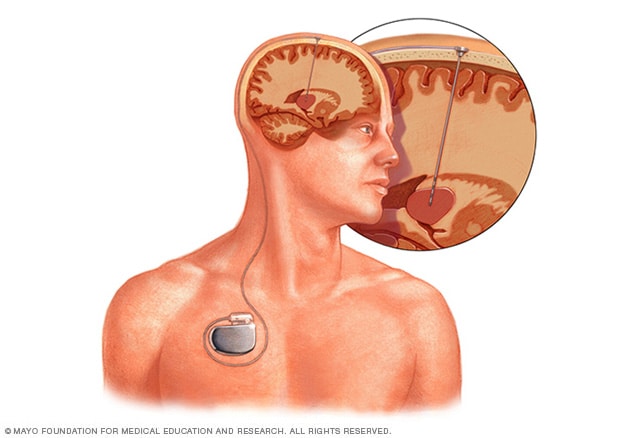
Deep brain stimulation
Deep brain stimulation involves implanting an electrode deep within the brain. The amount of electrical activity delivered by the electrode is controlled by a pacemakerlike device placed under the skin of the chest. A wire that travels under the skin connects the device to the electrode.
In general, here's how surgery for deep brain stimulation works:
-
Brain surgery. For the brain surgery, your healthcare team fits you with a special head frame called a stereotactic head frame. The frame keeps your head still during the procedure. Then, imaging such as brain MRI or CT is used to map your brain. The imaging pinpoints the proper areas in your brain to place the electrodes.
Most often, the electrodes are placed while you're awake and alert. This is to be sure the effects of deep brain stimulation can be tested fully. If you're awake for surgery, you'll likely be given a local anesthetic to numb your scalp before the procedure. You won't need an anesthetic in your brain itself because the brain has no pain receptors. In some people, surgery can be done using a medicine that puts you to sleep, known as general anesthetic.
Your surgeon implants a thin wire lead with several electrodes at the tips. The lead is placed into a specific area of your brain. Some people have leads implanted into each side of the brain for a total of two leads. A wire runs under your skin to a device called a pulse generator implanted near your collarbone.
During surgery, both the neurologist and the surgeon carefully monitor your brain to help ensure correct electrode placement.
-
Chest wall surgery. During the second part of the surgery, the surgeon implants the pulse generator under the skin of the chest, near the collarbone. This part of the device contains the batteries.
A medicine that puts you to sleep, known as general anesthesia, is used during this procedure. Wires from the brain electrodes are placed under your skin and guided down to the battery-operated pulse generator.
Several weeks after surgery, your healthcare professional programs the generator during an appointment. After it's programmed, the generator sends continuous electrical pulses to your brain. You control the generator and can turn it on or off using a remote control.
After the procedure
A few weeks after surgery, the pulse generator in your chest is activated, often in your healthcare professional's office. The pulse generator can be programmed from outside your body using a special remote control that you take home. The amount of electrical activity you use is customized to your condition. It may take as long as 4 to 6 months to find the best setting.
The pulse generator may be turned on 24 hours a day, or your care team may tell you to turn it off at night and back on in the morning. Sometimes the pulse generator may be programmed to let you make minor changes at home.
The battery life of the pulse generator varies with how often it's used and its settings. When the battery needs to be replaced, your surgeon replaces the generator during an outpatient procedure.
Results
Deep brain stimulation won't cure your condition, but it may help lessen your symptoms. While your symptoms may improve enough to make a difference, they often don't go away and you may need to continue taking medicines.
Deep brain stimulation isn't successful for everyone. Its success relies on several factors. Talk with a healthcare professional before surgery about what type of improvement you can expect.
Clinical trials
Explore Mayo Clinic studies of tests and procedures to help prevent, detect, treat or manage conditions.