Jan. 25, 2022
Capsule endoscopy (CE) is emerging as an effective tool for the diagnosis and management of inflammatory bowel disease (IBD). In an article published in Gastrointestinal Endoscopy Clinics of North America in 2021, co-authors Josiah D. McCain, M.D., Shabana F. Pasha, M.D., and Jonathan A. Leighton, M.D., discuss the indications, strengths and limitations of CE in the diagnosis and management of IBD, as well as the current trends and future directions for the use of this tool.
Drs. Pasha and Leighton are gastroenterologists at Mayo Clinic in Scottsdale, Arizona, and Dr. McCain is a Mayo Clinic gastroenterology fellow. In this Q and A, the co-authors answer key questions related to the use of CE in patients with IBD.
What are some benefits associated with CE when compared with traditional endoscopy in patients with Crohn's disease?
Changes consistent with Crohn's disease
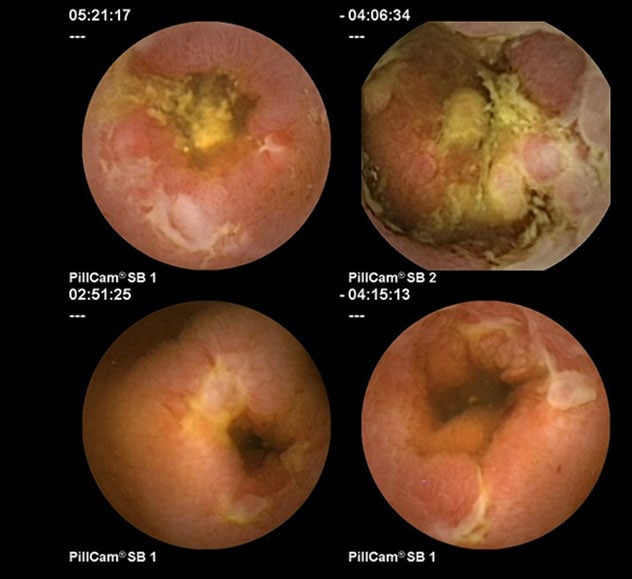
Changes consistent with Crohn's disease
Capsule endoscopy images of the small bowel show changes consistent with Crohn's disease.
Changes consistent with NSAID enteropathy
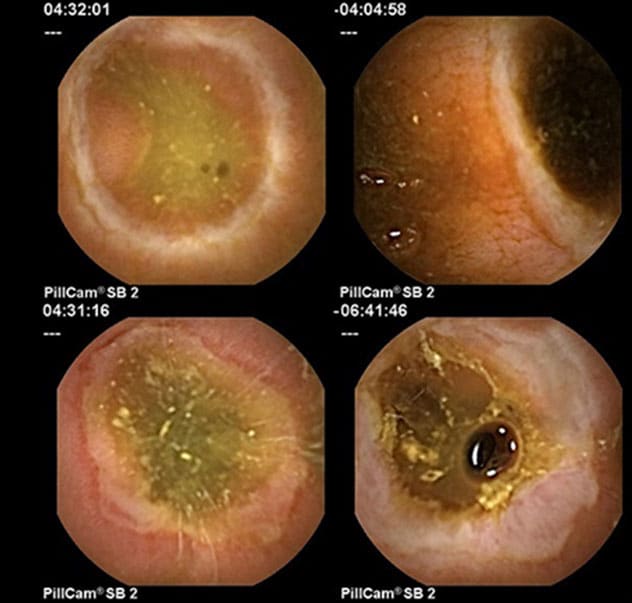
Changes consistent with NSAID enteropathy
Capsule endoscopy images of the small bowel show changes consistent with NSAID enteropathy.
Small bowel CE provides us with a well-tolerated, less invasive way to visualize the entire length of the small bowel in patients with Crohn's disease (CD). In most patients with suspected CD, ileocolonoscopy is still the most appropriate first evaluation. However, small bowel CE can be useful when ileocolonoscopy is negative, especially if small bowel cross-sectional imaging is also negative, and the index of suspicion for CD remains high. In patients with established CD, CE can help determine extent of disease, assess its severity in the small bowel and assist in monitoring response to therapy.
Can you explain CE's role in assessing mucosal healing and treat-to-target progress in patients with established CD?
Clinical symptoms do not consistently correlate with disease activity, and therefore endoscopic visualization of the mucosa is the most objective way to evaluate and document response to medical therapy in patients with CD. Mucosal healing in the small bowel after initiation of medical management is predictive of reduced activity in the future and overall better patient outcomes. Because CE can detect mucosal inflammation that might be missed by less sensitive modalities, it facilitates monitoring of disease activity more accurately, and it can thus guide management.
Cross-sectional studies such as computerized tomography (CT) or magnetic resonance (MR) enterography are also helpful to evaluate response to therapy. But CE may have a role in monitoring select patients with nonstricturing small bowel CD to confirm mucosal healing. Pan-enteric capsule endoscopy (PCE), in particular, might have a potential role in the future for assessment of patients with small bowel and colonic CD.
What role can CE play in diagnosing and managing UC?
While small bowel CE has been fairly well studied in CD, colon capsule endoscopy (CCE) for ulcerative colitis (UC) has not been as well studied. Therefore, the published evidence demonstrating the use of CCE in individuals with UC is still fairly limited. Small bowel CE is useful in ruling out small bowel CD in patients with IBD when there is not a clear diagnosis of UC. Ileocolonoscopy is currently the diagnostic procedure of choice in patients with UC. Colon capsule endoscopy (CCE) has potential as a noninvasive tool for diagnosis and monitoring of UC.
In our article we share available data from the literature showing that CCE has a fairly high degree of specificity and sensitivity, and substantial agreement with colonoscopy findings using the Mayo endoscopic score and the Ulcerative Colitis Endoscopic Index of Severity. In addition, CCE was both better tolerated and preferred by patients over colonoscopy. Further studies are needed to evaluate this role for CCE.
What more do we need to learn about using this tool in managing UC?
The precise role of CCE in UC remains to be defined. More research needs to be performed before we can determine the true diagnostic accuracy of this test in patients with this disease.
If mucosal healing remains a primary endpoint of UC management, patients will need to undergo periodic endoscopic surveillance in addition to other noninvasive tests such as fecal calprotectin. CCE's advantages for this application include its tolerability and less invasive nature, its sensitivity, and the fact it requires less time away from work for patients. It's important to acknowledge that CCE requires patients to undergo an extensive bowel prep with boosters. And unlike ileocolonoscopy, it does not allow for tissue sampling. We also need more data on the cost-effectiveness of CCE compared with ileocolonoscopy.
How significant is the risk of capsule retention?
In general, we have good evidence that CE is a very low-risk procedure with one major exception: potential retention of the video capsule. The risk of capsule retention in the general population is probably very low and estimates of its incidence range from 1.0% to 2.5%. Capsule retention rates are higher (2.6%) when evaluating suspected CD.
In patients with established CD, risk of capsule retention is much higher as patients may have strictures even in the absence of obstructive symptoms. It is recommended that patients with established CD be pre-screened to minimize risk of capsule retention. The risk can be significantly reduced by performing patency capsule or cross-sectional small bowel imaging prior to CE.
Can you share guidelines for management of capsule retention?
We recommend a conservative, observational approach as the first treatment for capsule retention, as many patients will pass the capsule spontaneously. If needed, medical therapy such as steroids or biologic agents can be helpful, especially for patients with Crohn's disease. Endoscopic or surgical retrieval of a capsule should only be performed in patients in whom observation and medical therapy have failed.
For more information
McCain JD, et al. Role of capsule endoscopy in inflammatory bowel disease. Gastrointestinal Endoscopy Clinics of North America. 2021;31:345.
Refer a patient to Mayo Clinic.