Overview
Coronary angioplasty (AN-jee-o-plas-tee) is a treatment to open clogged blood vessels of the heart. A tiny balloon on a narrow tube, called a catheter, is used to widen a clogged artery. This helps improve blood flow.
Angioplasty often is followed by the placement of a small wire mesh tube called a stent. The stent helps prop the artery open and lowers the chance of the artery narrowing again. Most stents are coated with medicine that helps keep the artery open.
Angioplasty with stent placement may be a planned treatment to improve blood flow to the heart muscles. Or it may be done as an emergency treatment for a heart attack.
Coronary angioplasty with stent placement also is called percutaneous coronary intervention.
Products & Services
Why it's done
Development of atherosclerosis
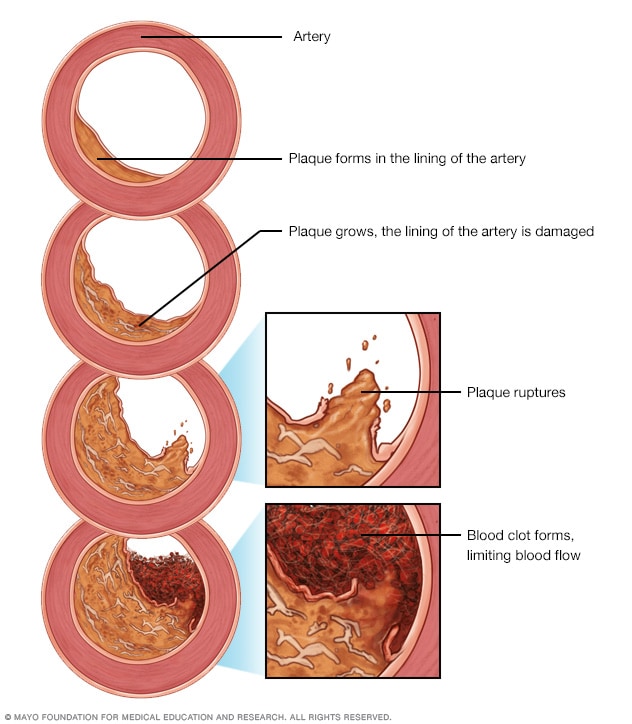
Development of atherosclerosis
If there's too much cholesterol in the blood, the cholesterol and other substances may form deposits called plaque. Plaque can cause an artery to become narrowed or blocked. If a plaque ruptures, a blood clot can form. Plaque and blood clots can reduce blood flow through an artery.
Angioplasty with stent placement is used to treat the buildup of fats, cholesterol, and other substances in and on the artery walls, a condition called atherosclerosis. Atherosclerosis is a common cause of blockages in the heart arteries. Blockage or narrowing of these blood vessels is called coronary artery disease.
Angioplasty improves blood flow to the heart. Your healthcare team may recommend this treatment if:
- Medicines or lifestyle changes have not improved heart health.
- Chest pain, called angina, caused by blocked arteries is becoming worse.
- Blood flow needs to be fixed quickly to treat a heart attack.
Angioplasty isn't for everyone. Sometimes an open-heart surgery called coronary artery bypass grafting is recommended instead. Another name for this surgery is CABG — pronounced "cabbage." It creates a new path for blood to flow around a blocked or partially blocked artery in the heart.
A heart doctor, called a cardiologist, and other members of your care team consider the severity of your heart disease and your overall health when deciding on the best treatment option.
Risks
Risks of coronary angioplasty with stent placement may include:
- Re-narrowing of the artery. Re-narrowing of the artery, also called re-stenosis, is more likely to happen if no stent is used. If the stent is coated with a medicine, there is even less risk of narrowing.
- Blood clots. Blood clots can form within stents. These clots can close the artery, causing a heart attack. Medicines can help lower the risk of blood clots.
- Bleeding or infection. During the angioplasty, a catheter is placed into a blood vessel, usually in the arm or leg. Bleeding, bruising or infection may happen where the catheter was placed.
Other rare risks of angioplasty include:
- Heart attack. Heart attacks that cause severe tissue damage or death are rare.
- Coronary artery damage. The coronary artery may be torn or broken open during coronary angioplasty and stenting. These complications may require emergency open-heart surgery.
- Kidney injury. The risk is higher when other conditions already affect how well the kidneys work.
- Stroke. During angioplasty, a piece of fatty plaque can break loose, travel to the brain and block blood flow. A stroke is an extremely rare complication of coronary angioplasty. Blood thinners are used during the treatment to lower the risk.
- Irregular heartbeats. During the treatment, the heart may beat too fast or too slow. These heart rhythm problems may need to be treated with medicine or a temporary pacemaker.
How you prepare
There may not be time to prepare. Sometimes, coronary angioplasty and stent placement are not scheduled. They may be done as emergency treatments for a heart attack.
If the treatment is scheduled, you can take some steps to prepare. A heart doctor called a cardiologist examines you and reviews your medical history. Tests are done to check your heart health and other conditions that may raise the risk of complications.
Your healthcare team gives you instructions to help you prepare. You may be asked to do the following:
- Write down all medicines, dietary supplements and herbal treatments you take. Include dosages.
- Adjust or stop taking certain medicines before angioplasty, such as aspirin, nonsteroidal anti-inflammatory drugs known as NSAIDs or blood thinners. Ask your healthcare team which medicines you need to stop taking and which medicines you need to keep taking.
- Do not eat or drink for several hours before your treatment.
- Take approved medicines with small sips of water on the morning of your treatment.
- Arrange for a ride home.
What you can expect
Before
Sticky patches called electrodes are placed on your chest to check your heartbeat. Your blood pressure also is checked. Your heart rate, pulse, blood pressure and oxygen level are continuously checked before, during and after the treatment.
A member of your healthcare team may shave any hair from the body area where a flexible tube called a catheter will be inserted.
During
Coronary artery stent
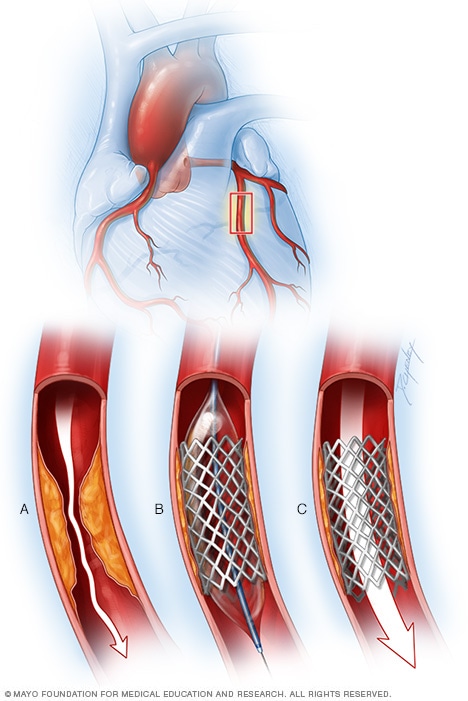
Coronary artery stent
To place a coronary artery stent, a balloon on the tip of the catheter is inflated to widen the blocked artery (A). Then a metal mesh stent is placed (B). The stent helps hold the artery open so blood can flow through it (C).
Coronary angioplasty with stenting usually is done in the hospital. The hospital room has special X-ray and imaging machines.
A healthcare professional places an IV into your arm or hand. You get medicines, called sedatives, to help you relax. The amount of sedation needed for coronary angioplasty and stenting depends on your health and why you're having the treatment. Usually you are awake during the treatment. But some people may get a combination of medicines that puts them to sleep during the treatment. This is called general anesthesia.
Fluids and other medicines, such as blood thinners, also are given through the IV.
The area where the catheter goes is numbed. Then, the doctor makes a small cut to reach the blood vessel. The catheter is placed into the blood vessel and guided to the heart.
Dye flows through the catheter. The dye helps blood vessels show up more clearly on the X-ray images. Doing this helps the doctor check for blockages in the heart arteries. The results help your doctor decide whether to continue with the angioplasty.
Angioplasty and stent placement
Angioplasty is the process of opening an artery by inflating a balloon. A stent is a mesh coil that helps hold the artery open. Most stents slowly release a medicine that prevents the artery from re-narrowing.
During angioplasty and stenting, flexible tubes called catheters and a balloon are used to reopen a blocked artery. A catheter with an empty balloon on the tip is guided to the blocked artery. The balloon inflates, widening the artery. The balloon is deflated and removed. A stent is placed in the narrowed part to help keep the artery open.
If there's more than one blockage, the process may be repeated.
You might feel pressure in the area where the catheter is placed. You also may feel some mild discomfort when the balloon is inflated.
Your healthcare team tells you how long the treatment is expected to take. The treatment and recovery typically takes several hours.
After
How long you stay in the hospital after angioplasty and stenting depends on why the treatment was done.
When you get home, rest and drink plenty of fluids to help remove the imaging dye from your body. You can expect the following after coronary angioplasty and stenting:
- Blood-thinning medicines. After getting a stent, you may need to take medicines to prevent blood clots. Your healthcare professional may recommend taking aspirin with another medicine, such as clopidogrel (Plavix), ticagrelor (Brilinta) or prasugrel (Effient). Aspirin recommendations vary. Check with your healthcare team before starting aspirin.
- Activity restrictions. Do not do strenuous exercise or lift heavy objects for at least 24 hours after coronary angioplasty and stenting. Ask your healthcare team if you have any other activity restrictions.
- Cardiac rehabilitation. Your heart doctor may suggest a personalized program of exercise and education called cardiac rehabilitation. It typically involves exercise training, emotional support and education about a heart-healthy lifestyle. The supervised program is designed to improve health in those with heart disease. It's often recommended after a heart attack or heart surgery.
After coronary angioplasty and stenting, it's important to watch for complications. Call your healthcare professional's office or hospital team right away if you have:
- Chest pain or shortness of breath.
- Weakness or fainting.
- Bleeding or swelling at the body area where the catheter was placed.
- Pain or discomfort near the area where the catheter was placed.
- Symptoms of infection, such as a change in skin color, swelling, drainage or fever.
- A change in temperature or color of the leg or arm that was used for the treatment.
You need regular health checkups after coronary angioplasty and stenting. During these checkups, blood and imaging tests may be done to see how well your heart is working.
Results
Coronary angioplasty and stent placement can greatly increase blood flow through a heart artery that used to be blocked or narrowed. Your doctor can compare images of your heart taken before and after the treatment to find out how well the angioplasty and stenting has worked.
Angioplasty with stenting does not treat the underlying causes of blockages in your arteries. To keep your heart healthy after angioplasty, try these tips:
- Do not smoke or use tobacco.
- Eat a diet that is low in saturated fats and rich in vegetables, fruits, whole grains, and healthy oils such as olive oil or avocado oil.
- Stay at a healthy weight. Ask a healthcare professional what a healthy weight is for you.
- Get regular exercise.
- Control cholesterol, blood pressure and blood sugar.