Overview
Mirena is brand name for a hormonal intrauterine device (IUD). A hormonal IUD is a type of birth control that's placed in the uterus and uses hormones to give long-term birth control. Birth control also is called contraception.
The device is a T-shaped plastic frame. It releases a type of the hormone progestin.
To prevent pregnancy, Mirena:
- Thickens mucus in the cervix to stop sperm from reaching an egg or making it fertile.
- Thins the lining of the uterus and partly stops the release of an egg. The release of the egg is called ovulation.
Mirena is one of several hormonal IUDs that the Food and Drug Administration has approved. It's been shown to prevent pregnancy for up to eight years.
Why it's done
Mirena gives long-term birth control. It can be used in people assigned female at birth who haven't been through menopause. This includes teenagers.
Among various pluses, Mirena:
- Is always ready for use, so you don't need to stop in the middle of sex for birth control.
- Doesn't need your partner to be involved with birth control.
- Can stay in place for eight years.
- Can be removed at any time. And you can get pregnant soon after it's out.
- Can be used while breastfeeding. However, the risk of tearing the uterus while placing the IUD is higher in people who are breastfeeding.
- Can be used for emergency birth control if put in within five days of having sex with no protection.
After three or more months of using Mirena, you might bleed less during your period. About 20% of people who use Mirena stop having periods after one year of use.
Mirena also can lessen:
- Bad menstrual pain and pain related to the growth of uterine-lining tissue outside the uterus, called endometriosis.
- The risk of pelvic infection.
- The risk of cancer of the lining of the uterus, called endometrial cancer.
Because of these benefits, Mirena often is prescribed for people with:
- Heavy bleeding during periods.
- Cramping or pain with periods.
- Endometriosis.
- Unusual growth of the lining of the uterus, called endometrial hyperplasia.
- Unusual growth of uterine-lining tissue into the muscles of the wall of the uterus, called adenomyosis.
- A lack of enough healthy red blood cells to carry oxygen through the body, called anemia.
- Growths in the uterus that aren't cancer, called fibroids.
Mirena isn't right for everyone. Your healthcare professional might not want to prescribe Mirena if you have:
- Breast cancer or have had it.
- Uterine or cervical cancer.
- Liver disease.
- Uterine conditions, such as fibroids, that can cause problems with placing Mirena or keeping it in.
- A pelvic infection or current pelvic inflammatory disease.
- Vaginal bleeding with no known cause.
- A uterus that is not formed typically.
Tell your healthcare professional if you:
- Take any medicines, including herbal products, supplements and medicines available without a prescription.
- Have diabetes or high blood pressure.
- Have a heart condition or have had a heart attack.
- Have migraines.
- Have blood-clotting problems or have had a stroke.
- Recently gave birth or are breastfeeding.
Risks
Less than 1% of people who use Mirena will get pregnant in a year of typical use.
If you do get pregnant while using Mirena, you're at higher risk of having the egg implant outside the uterus. That is called an ectopic pregnancy. In ectopic pregnancies, the egg most often is in a fallopian tube. But because Mirena prevents most pregnancies, people who use it are at lower risk of having an ectopic pregnancy than are others who have sex without using birth control.
Mirena is mainly safe. But know that:
- Mirena doesn't protect against sexually transmitted infections, called STIs.
- Rarely, putting in Mirena causes a tear in the uterus. The risk of tearing might be higher when Mirena is put in right after giving birth.
Side effects linked to Mirena include:
- Headache.
- Acne.
- Breast soreness.
- Irregular bleeding. This can get better after three months of use.
- Mood changes.
- Cramping or pelvic pain.
Mirena also can come out of the uterus. This may be more likely to happen if you:
- Have never been pregnant.
- Have heavy or long periods.
- Have bad menstrual pain.
- Had an IUD come out on its own before.
- Are younger than age 20.
- Had Mirena put in right after childbirth.
Your healthcare professional may suggest removing Mirena if you get:
- A pelvic infection. But some infections can be treated with the IUD in place.
- A condition that causes swelling and irritation of the lining of the uterus, called endometritis.
- Endometrial or cervical cancer.
- Pelvic pain or pain during sex.
- Very painful migraines.
- A big increase in blood pressure or have a stroke or heart attack.
How you prepare
Your healthcare professional will check your health and do a pelvic exam before putting in Mirena. You may be screened for STIs.
Mirena can be put in:
- During your menstrual cycle if you're not pregnant. You might need to take a pregnancy test to make sure you're not pregnant.
- Right after ending a pregnancy.
- Right after giving birth vaginally or by cesarean section. But putting in Mirena right after vaginal delivery raises the risk of Mirena coming out.
If you have Mirena put in more than seven days after the start of your period, be sure to use other birth control for one week.
Taking a nonsteroidal anti-inflammatory medicine, such as ibuprofen (Advil, Motrin IB, others), 1 to 2 hours before the procedure might help reduce cramping.
What you can expect
Mirena placement
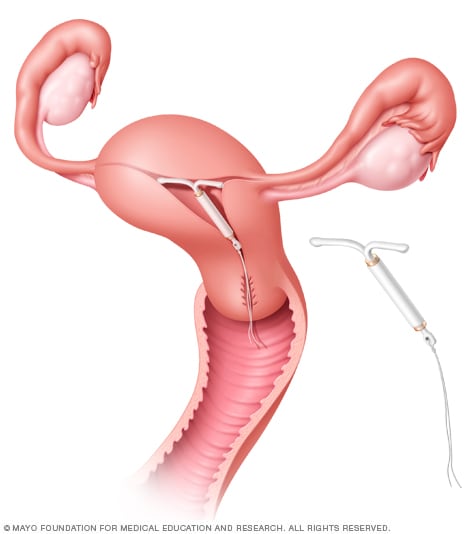
Mirena placement
The Mirena intrauterine device (IUD) is put into the uterus by a healthcare professional using a special applicator. Short strings attached to the device hang down into the vagina. The strings allow for taking the device out.
Mirena most often is put in at a healthcare professional's office.
During the procedure
Your healthcare professional puts a device called a speculum into your vagina to hold it open. The next step might be to clean your vagina and cervix with an antiseptic liquid that kills germs.
Special instruments might be used to hold the cervix still and line it up with the uterus. Your health professional looks at the size and shape of the uterus and measures how deep it is.
Next, your healthcare professional folds Mirena's arms and puts the device inside a tube that applies the IUD. The tube goes into your cervical canal to place Mirena in your uterus. Then the tube is removed and Mirena stays in place.
Your healthcare professional trims Mirena's strings so that they don't go too far into the vagina. You might be asked to feel the cut strings. That's so you know what to feel for when checking the strings.
During the procedure, you may cramp, feel dizzy or faint. Your heart rate might be slower than usual.
After the procedure
About a month after you get Mirena, your healthcare professional may do another pelvic exam. This is to make sure Mirena hasn't moved and to check for infection.
Some health professionals might ask you to check your vagina monthly to feel that Mirena's strings are still there.
While using Mirena, contact your health professional right away if you:
- Think you may be pregnant.
- Have ongoing vaginal bleeding that's heavier than usual.
- Have pelvic pain or pain during sex.
- Get a fever.
- Have vaginal sores or discharge that's not usual or that smells bad.
- Get very bad headaches or migraines.
- Have yellowing of the skin and yellowing of the eyes. Yellowing of the skin might be harder to see on some skin colors.
- Might have an STI. Most often the IUD can be left in place. But use condoms until the infection clears up.
- Can no longer feel the IUD strings or they feel longer than they were.
- Can feel part of the IUD, not just the strings.
Also contact your healthcare professional right away if you think Mirena is no longer in place. Your health professional will check for Mirena. If it's out of place, it might need to be taken out.
Removal
Mirena can stay in place for up to eight years. To remove Mirena, your healthcare professional uses a gloved hand or forceps to grasp the device's strings and gently pull. The device's arms will fold upward as it leaves the uterus.
Light bleeding and cramping are common during removal. Rarely, removal can be harder to do.