Oct. 23, 2014
Opciones endovasculares para la incorporación de bifurcaciones
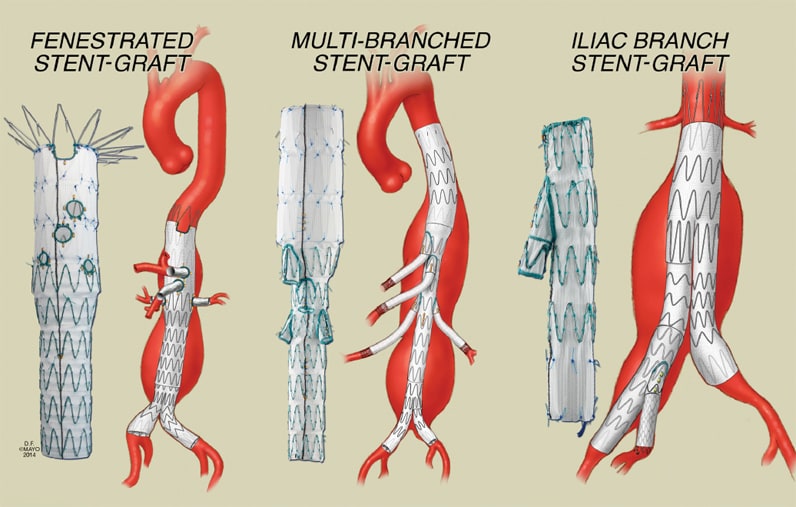
Opciones endovasculares para la incorporación de bifurcaciones
Las opciones endovasculares para la incorporación de bifurcaciones incluyen los dispositivos fenestrados y bifurcados para aneurismas pararrenales, los dispositivos multibifurcados para aneurismas toracoabdominales y los dispositivos de arteria ilíaca para aneurismas aortoilíacos.
Endovascular aortic aneurysm repair (EVAR) has been shown to reduce blood loss, operative time, length of hospital stay, mortality, and morbidity compared with open surgical repair of infrarenal abdominal aortic aneurysms (AAAs). Anatomical constraints limit the use of EVAR in 30 to 40 percent of patients, because of short necks, excessive angulation, or frank aneurysm involvement of aortic side branches such as supra-aortic trunks (arch aneurysms), visceral arteries (thoracoabdominal and pararenal aneurysms), or the internal iliac arteries.
"Fenestrated and branched stent-grafts allow incorporation of aortic side branches and preservation of end-organ perfusion while achieving aneurysm exclusion with a total endovascular approach," says Gustavo S. Oderich, M.D., director of endovascular therapy and a vascular surgeon at Mayo Clinic's campus in Rochester, Minnesota. Single-center reports, multicenter registries, and systematic reviews indicate that the technique is reproducible with high rates of technical success and low morbidity and mortality.
Open conventional repair remains the standard treatment for patients with complex aortic aneurysms, but this necessitates extensive dissection, higher clamp site, prolonged visceral ischemia and more extensive reconstruction. While contemporary series have shown that open repair of thoracoabdominal aortic aneurysms (TAAAs) can be performed with mortality rates in the range of 5 to 15 percent in high-volume centers, analyses of national and regional data sets have demonstrated more-ominous results. In the study by Rigberg and associates on 797 Medicare beneficiaries who underwent elective open TAAA repair in California, the mortality rate was 19 percent at 30 days and 31 percent at 1 year.
Dr. Oderich and colleagues at Mayo Clinic have reported the results of 5,798 Medicare patients who underwent open repair in the United States. Mortality increased from 15 percent for patients ages 60 to 70 years to 30 percent for those older than 80 years. Overall, a third of the patients were dismissed home and the remainder required prolonged recovery in rehabilitation centers or nursing homes.
Endovascular strategies
The initial experiences with fenestrated and branched endografts have shown that total endovascular repair is effective and may reduce morbidity rates in patients with arch, thoracoabdominal and pararenal aneurysms. Most of these devices are not yet available for commercial use in the United States and require a period of customization of six to eight weeks.
In the absence of widely available endograft designs, a number of centers have reported on a hybrid technique that applies surgical bypasses to reroute the visceral arteries while endovascular stent-grafts are placed to exclude the aneurysm. While this procedure avoids thoracotomy and in many patients aortic cross-clamping, enthusiasm with early results has been tempered by high morbidity and mortality in most reports. Currently, most centers with access to fenestrated and branched endografts have relegated hybrid procedures to high-risk patients who are candidates for neither total endovascular nor open surgical repair.
Definitions
Aortic side branches can be incorporated into stent-grafts using either fenestrations or directional branches. Fenestrations imply side holes in the fabric of the stent-graft. These are reinforced by a nitinol ring to facilitate catheterization, prevent fraying of the fabric, and allow attachment of a side branch alignment stent.
The term "fenestrated endovascular repair" is applied when a fenestrated stent-graft is used to repair an aneurysm with an inadequate or short infrarenal neck, yet the target vessels (for example, renal arteries) arise from a normal segment of the aorta. There is no gap between the fenestration and the target vessel; an alignment stent is typically inserted into side branches to prevent vessel occlusion or stenosis from misalignment between the fenestration and the origin of the target vessel.
Directional or cuffed branches imply the presence of presewn cuffs in the main aortic stent-graft, which serve as a docking site for placement of stent-grafts connecting the main aortic component into each of the side branches. Whereas fenestrations are usually connected with balloon-expandable stents, branches are bridged by self-expandable stent-grafts.
Branched endovascular repair is the term used to describe endovascular repair of aneurysms with involvement of side branches. The target vessel originates from the aneurysm, and there is a gap between the main stent-graft and the aortic wall.
Availability of devices in the U.S.
Current designs allow incorporation of almost any large-diameter aortic side branch, including aortic arch, visceral, and internal iliac arteries. In the United States, access to fenestrated and branched stent-grafts is limited to a few institutions.
The Zenith fenestrated stent-graft (Cook Medical Inc) was approved for commercial use by the Food and Drug Administration in April 2012. This device is indicated for short-neck infrarenal or juxtarenal aneurysms, allowing a maximum of three custom-made fenestrations built into the device. While this represents a significant advance, patients with more-complex aneurysms, such as those with pararenal, paravisceral, or thoracoabdominal aneurysms, are not candidates for this device. In these patients, fenestrated and branched stent-grafts are available under physician-sponsored investigational device exemption (PS-IDE) protocols or industry-sponsored trials.
Preoperative evaluation
A comprehensive evaluation of cardiac, pulmonary, and renal performance is a crucial component in optimal patient selection. These procedures are often indicated in the sickest patients, but clinical data suggests that benefits may be higher in low- or intermediate-risk patients. Prohibitively high-risk patients and those with limited life expectancy are not ideal candidates. The evaluation typically includes a noninvasive cardiac stress test, pulmonary function tests, and a carotid ultrasound.
Design and planning is based on careful analysis of aneurysm morphology using high-resolution computerized tomography angiography (CTA) with small (1- to 3-mm) cuts. Standard measurements include centerline of flow analysis to determine accurate estimates of lengths, axial clock position, arc lengths and angles. Device planning starts with the selection of an adequate proximal landing zone based on a "healthy" aorta. A normal aorta should have parallel walls with no calcium or thrombus. In patients with diffuse aortic ectasia, familial history of aortic aneurysm, or minor aortic abnormalities (calcium or thrombus), a generous proximal landing zone of 6 to 8 cm is recommended above the level of the celiac axis.
Juxtarenal or pararenal aortic aneurysms
Aneurysms involving the renal arteries but not extending beyond the superior mesenteric artery are repaired using fenestrated stent-grafts. The Zenith fenestrated stent-graft has been approved for treatment of patients with > 4 mm neck below the renal arteries, but these devices can be fashioned with more fenestrations in centers with PS-IDE protocols. Based on contemporary results, technical success is greater than 99 percent, with average 30-day mortality of 1.5 percent (0 percent-4 percent).
Attachment endoleaks are infrequent, occurring in less than 2 percent of patients, but approximately 15 to 20 percent may have a retrograde endoleak via the lumbar arteries. These are typically observed or treated by percutaneous coil embolization if there is evidence of aneurysm sac growth. Patency of visceral branches is exceptionally high, averaging > 95 percent at five years. Secondary intervention rates range between 10 and 20 percent. Modest deterioration of renal function is not uncommon (up to 25 percent), though incidence of dialysis is low (0 to 3 percent). Three systematic reviews of the literature have shown that fenestrated repair is associated with significantly lower rates of early mortality and renal dysfunction as compared with open surgical repair.
Thoracoabdominal aortic aneurysms
Tratamiento del aneurisma toracoabdominal extenso después de la reparación previa de la aorta infrarrenal
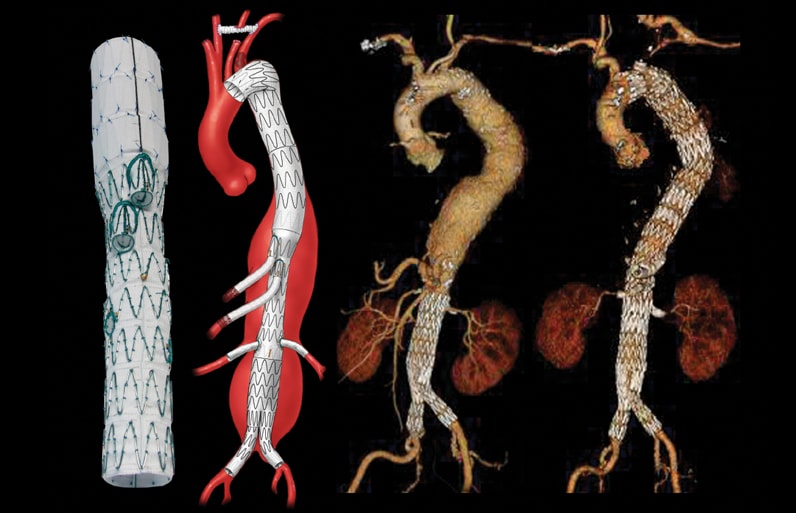
Tratamiento del aneurisma toracoabdominal extenso después de la reparación previa de la aorta infrarrenal
El aneurisma toracoabdominal extenso después de la reparación previa de la aorta infrarrenal se trató mediante un endoinjerto ramificado con dos ramas en el caso de las arterias mesentéricas superior y celíaca, y fenestraciones en el caso de las arterias renales.
Thoracoabdominal aneurysms extend across the visceral arteries and represent the most complex form of aortic aneurysm. Technical difficulty is determined by extent of aortic involvement, visceral occlusive disease, tortuosity, and iliac access issues:
- Type I and Type II TAAAs involve the entire thoracic aorta extending distally to or beyond the renal arteries, respectively.
- Type III TAAAs start below the T6 level and extend beyond the renal arteries.
- Type IV TAAAs are the simplest form, starting at the level of the celiac axis and extending distal into the infrarenal aorta or iliac arteries.
The extent of aortic involvement has direct implications for choice of technique (fenestrated or branched) and risk of spinal cord injury, which ranges from 1 to 2 percent for Type IV TAAAs and up to 10 percent for Type II TAAAs.
Device design can be done with fenestrations, branches, or both. Each design has its own advantages and disadvantages. Fenestrations are optimal for renal arteries and for vessels that originate from a narrow aortic lumen, while branches are ideal for the celiac and superior mesenteric arteries or for branches that originate from large aortic lumen.
Contemporary reports have shown that branched stent-grafts can be performed with high technical success (93 to 100 percent). Mortality averages 4 percent in systematic reviews, ranging from 0 to 18 percent in single-center reports. Incidence of dialysis averages 2 percent, ranging from 0 to 8 percent. Spinal cord injury has been improved by staged repair, which allows conditioning of spinal cord collaterals with paraplegia rates of 5 percent in the most extensive (Type II) TAAAs. Visceral branch patency is > 95 percent. Secondary interventions are required in 10 to 15 percent of patients.
Aortoiliac aneurysms
Approximately 30 percent of patients treated by EVAR have ectatic or aneurysmal common iliac arteries, which are not suitable to seal the stent-graft. In these patients, one of the most commonly utilized options is unilateral or bilateral internal iliac artery exclusion, allowing extension of the stent-graft into the external iliac artery.
Buttock claudication may occur in 16 to 50 percent of patients treated by unilateral and up to 80 percent of those undergoing bilateral embolization. Sexual dysfunction is noted in 10 to 17 percent of patients. Although uncommon, devastating complications include spinal cord injury, ischemic colitis, and gluteal muscle necrosis.
In addition, because the internal iliac artery is an important collateral to the spinal cord, preservation of internal iliac artery flow is critical to prevent paraplegia in patients with concomitant thoracic or thoracoabdominal aortic aneurysms. Results of iliac branch stent-grafts have been encouraging, with technical success rates of greater than 95 percent in most reports and branch patency rates of more than 95 percent at five years.
Summary
"Endovascular repair of complex aneurysms involving the visceral arteries has become a reality," says Dr. Oderich. "Fenestrated stent-grafts are increasingly utilized to treat pararenal and thoracoabdominal aneurysms. The technique is safe, effective, and can be performed with high technical success and low risk of complications by experienced physicians."
More than 8,000 patients have been treated by fenestrated and branched stent-grafts and more than 5,500 by iliac branch devices. Based on results of single-center reports, systematic reviews, and the U.S. prospective trial, technical success is high (> 98 percent) with low rates of type I and type III endoleak, migration, aneurysm rupture, and conversion to open repair. Branch patency averages > 95 percent with covered stents.
These results should serve as a benchmark for comparison with alternative endovascular techniques of branch vessel incorporation, including debranching, snorkel, and physician-modified grafts. "Long-term comparison with open surgical repair is still required," says Dr. Oderich.
Clinical trials
Clinical trials and physician-sponsored investigational device exemption (PS-IDE) protocols are available at Mayo Clinic to treat arch, thoracoabdominal, and pararenal aneurysms using manufactured fenestrated and branched stent-grafts.
For more information about clinical trials utilizing these devices in the treatment of aortic aneurysms at Mayo Clinic, contact Gustavo S. Oderich, M.D., at 507-284-1575 or oderich.gustavo@mayo.edu.