June 01, 2019
Artificial intelligence (AI), defined as intelligence exhibited by machines, conjures up images of HAL, the rogue computer in Stanley Kubrick's epic 1968 science fiction movie, "2001: A Space Odyssey." The idea of intelligence being incorporated into inanimate objects has been floated since antiquity. The first rudimentary AI programs were developed in 1951 to play checkers and chess. Since then, AI has expanded to almost every facet of modern life, including the medical field.
The collaborative Mayo Clinic cardiovascular AI team recently published the results of their study utilizing AI electrocardiogram (ECG) analysis to predict the presence of left ventricular dysfunction in asymptomatic patients. Study results were published in Nature Medicine in 2019.
Some basics
Some common examples of machines that utilize versions of AI include:
- iRobot Roomba, which vacuums the floor and can navigate around obstacles
- Mars rovers Spirit and Opportunity
- Siri, Apple's virtual assistant
These and most other devices utilize weak (narrow) AI. They operate within a restricted range of pre-defined functions to accomplish narrowly demarcated tasks.
Deep learning ― strong AI ― is one of a family of machine learning methods based on learning data set representations. Advances in computing power so that large amounts of data can be quickly analyzed have made the application of AI to huge, complex data sets feasible.
Deep learning architectures have been applied to diverse fields such as speech recognition, social network filtering, bioinformatics, drug design and medical image interpretation.
Deep neural systems comprise a series of layers:
- An input layer
- A cascade of processing units or hidden layers
- An output layer
循环神经网络

循环神经网络
循环神经网络通过将单个矢量输入置于一系列隐藏层中进行变换。每一层都由一组神经元(圆)组成。每层内的神经元并不与同层的其他神经元共享连接;但是,每个神经元与上一层的每个神经元都有连接:它们是完全连接的。最后,还有最后一个完全连接的层(输出层),代表神经网络预测。
Each of the layers comprises individual neurons that extract and transfer data in a hierarchical fashion into more composite representations. Data from one layer is processed and fed into the next layer in a recurrent neural network. Different types of neural networks have been developed; the type of neural network employed depends on the type and complexity of analysis being performed.
卷积神经网络
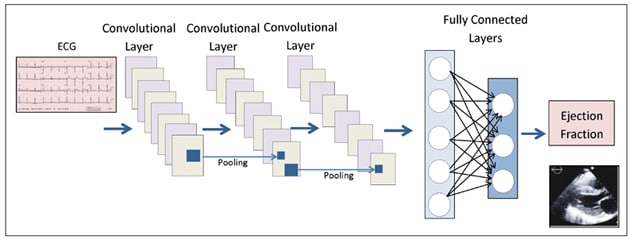
卷积神经网络
妙佑医疗国际心血管人工智能团队根据超声心动图(ECG)的细微发现预测无症状左心室功能障碍的卷积神经网络结构示意图。
池化示例
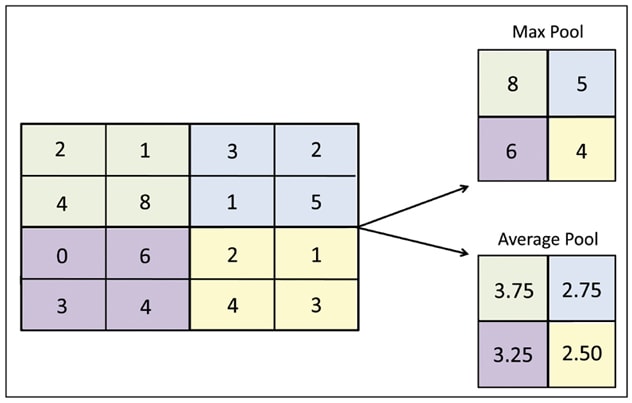
池化示例
关于原始矩阵输入(左)中出现的每一个颜色区域,右上方的过滤器会滤出该区域的最大值,并创建一个新的输出矩阵;这就是最常用的池化方法。右下方示例是举例说明如何用平均过滤器创建一个新的输出矩阵。
Because recurrent neural networks are not particularly good at image analysis, convolutional neural networks (CNNs) are commonly used for this function and are especially helpful in the evaluation of high-resolution medical imaging. Subtle variations in a data set can be identified, extracted and compiled. CNNs have two functional components:
- In feature extraction (convolutional layers), data from the input layer is fed into the hidden layers, which perform a series of convolutions, which are mathematical functions, and pooling operations which make assumptions about the data to downsize the number of parameters to analyze and neutralize the effect of changes in scale or orientation; computational cost is also reduced. Characteristic features in the image are detected during this process. For example, in a picture of a pig, this extraction process identifies four short legs with even-toed ungulate hooves, a curly tail, fat body, two pointy ears and a snout.
- In image classification (fully connected layers), the fully connected layers, in which each neuron in one layer is connected to every neuron in the adjacent layers, will classify the image based upon these extracted features. This process assigns a probability that the object in the image is actually what the algorithm predicts it is ― that is, a pig. Well-designed classification layers avoid considering broad areas of possibilities that are unlikely to be relevant. A pig lives on land; therefore, ocean dwellers need not be considered.
The study
Paul A. Friedman, M.D., electrophysiologist and chair of Cardiovascular Medicine at Mayo Clinic in Rochester, Minnesota, spearheaded the study utilizing CNNs to analyze ECG to predict the presence of asymptomatic left ventricular dysfunction (ALVD).
ALVD is present in 3% to 6% of the general population, affecting more than 7 million Americans. Risk increases with age; in the elderly, ALVD is present in 9% of individuals. Effective therapies exist and, if administered early, can reduce hospitalizations and mortality significantly.
Yet, no ALVD screening tool exists that is inexpensive, widely available and noninvasive to facilitate broad and early intervention by general clinicians. Currently, the gold standard screen for ALVD is echocardiogram, not readily available in nonspecialty clinics. B-type natriuretic peptide (BNP) levels have been studied for screening purposes, but results have been disappointing, and the test requires invasive phlebotomy.
Dr. Friedman and his team envisioned screening for ALVD by subjecting the common and inexpensive ECG to a custom-designed and trained CNN. Explains Dr. Friedman: "We hypothesized that the metabolic and structural derangements associated with the cardiomyopathic process would result in ECG changes that we could reliably detect with a properly trained neural network."
ECGs and echocardiographic images from Mayo Clinic's large digital database were used. From training inputs, the CNN generalized attributes to make predictions about images the network had never previously seen. Using Mayo Clinic stored digital data, researchers screened 625,326 paired ECG and transthoracic echocardiograms to identify the population to be studied for analysis. Because ECGs involve repeating complexes, both spatial and temporal dimensions were analyzed.
The team then trained and validated the CNN with patient data from 44,959 ECG-echocardiogram pairs. The network goal was to use ejection fractions (EFs) ≤ 35% to identify patients with low EFs and refer them for more detailed follow-up exams.
The team tested the network on 52,870 patients reserved for external validation. Of those, approximately 8% had an EF < 35%. The sensitivity, specificity and accuracy of the CNN were 85%, 86% and 86%, respectively. "The network ranked 0.93 in its ability to flag low EFs — out of a perfect score of 1.0. To put this in perspective, a mammogram is 0.85. These results show the network is very robust," Dr. Friedman says.
Additionally, when the network suggested a low EF based on ECG findings but the echo EF was normal, patients had an estimated fivefold increased risk of developing a low EF in the future. "This suggests that the network may reveal silent subclinical, metabolic or structural abnormalities hidden in the ECG," Dr. Friedman says. Within the next year, providers at Mayo Clinic will launch a pilot study of the use of AI to detect ALVD in the clinical practice.
Within Mayo Clinic Cardiovascular Medicine, interdisciplinary teams are applying artificial intelligence to some of the most challenging clinical problems. Exciting examples include:
- Early risk prediction of conditions such as embolic stroke
- Heart monitoring and arrhythmia detection in smart clothing projects based on a textile computing platform
- Occult disease detection, such as identifying atrial fibrillation's earliest, subclinical stages, through heart physiology signals transmitted by mobile ECG
For more information
Attia ZI, et al. Screening for cardiac contractile dysfunction using an artificial intelligence-enabled electrocardiogram. Nature Medicine. 2019;25:70.
For more information about AI cardiovascular research at Mayo Clinic, contact Francisco Lopez-Jimenez, M.D., at 507-284-8087.