Diagnosis
Pelvic exam
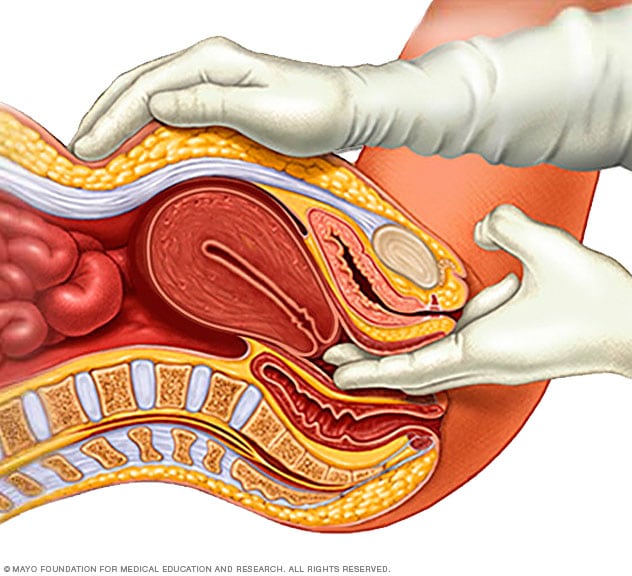
Pelvic exam
During a pelvic exam, a doctor inserts one or two gloved fingers inside the vagina. Pressing down on the abdomen at the same time, the doctor can check the uterus, ovaries and other organs.
Uterine fibroids often are found by chance during a routine pelvic exam. Your doctor may feel irregular changes in the shape of your uterus, suggesting the presence of fibroids.
If you have symptoms of uterine fibroids, you may need these tests:
-
Ultrasound. This test uses sound waves to get a picture of your uterus. It can confirm that you have fibroids, and map and measure them.
A doctor or technician moves the ultrasound device, called a transducer, over your stomach area. This is called a transabdominal ultrasound. Or the device is placed inside your vagina to get images of your uterus. This is called a transvaginal ultrasound.
- Lab tests. If you have irregular menstrual bleeding, you may need blood tests to look for possible causes of it. These might include a complete blood count to check for anemia due to ongoing blood loss. Other blood tests can search for bleeding disorders or thyroid problems.
Other imaging tests
Hysterosonography
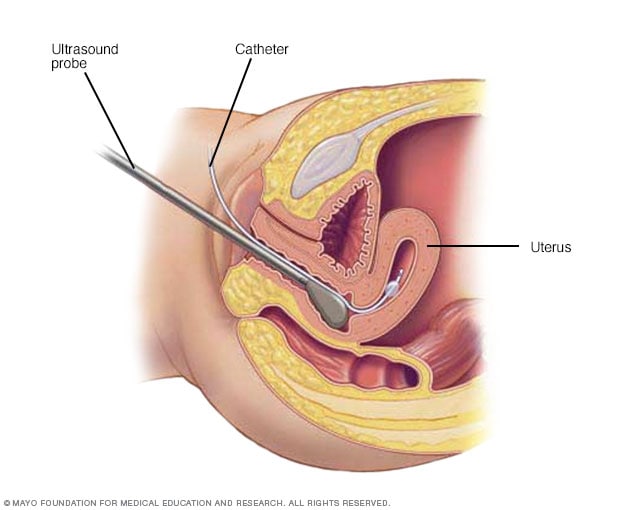
Hysterosonography
During hysterosonography (his-tur-o-suh-NOG-ruh-fee), you have a thin, flexible tube called a catheter placed in the uterus. Salt water, also called saline, is injected through the flexible tube into the hollow part of the uterus. An ultrasound probe transmits images of the inside of the uterus to a nearby monitor.
Hysterosalpingography
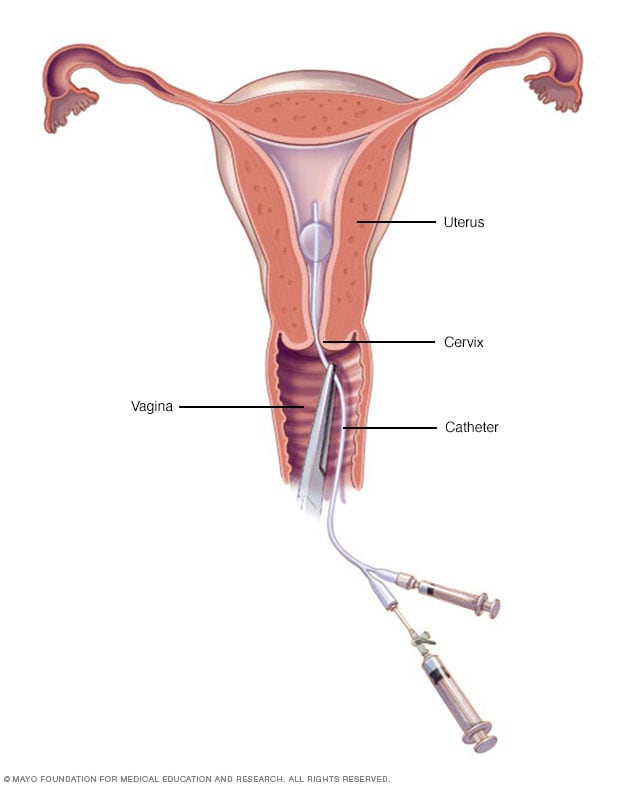
Hysterosalpingography
A doctor or technician places a slender catheter inside your cervix. It releases a liquid contrast material that flows into your uterus. The dye traces the shape of your uterine cavity and fallopian tubes and makes them visible on X-ray images.
Hysteroscopy
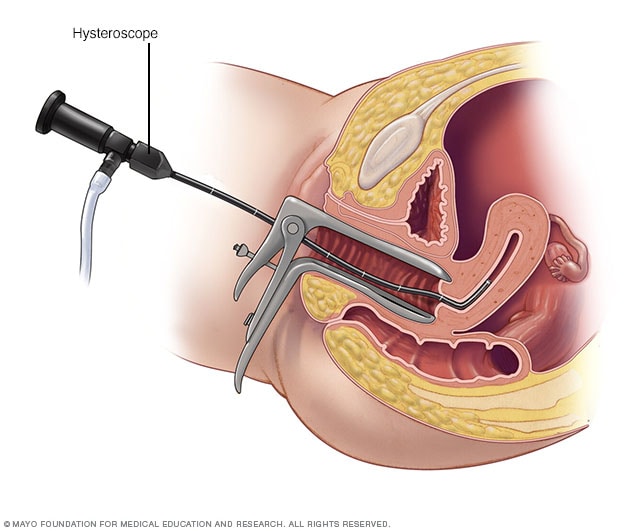
Hysteroscopy
During hysteroscopy (his-tur-OS-kuh-pee), a thin, lighted instrument provides a view of the inside of the uterus. This instrument also is called a hysteroscope.
If an ultrasound doesn't provide enough information, you may need other imaging studies, such as:
- Magnetic resonance imaging (MRI). This test can show in more detail the size and location of fibroids. It also can identify different types of tumors and help determine treatment options. Most often, an MRI is used in people with a larger uterus or in those who are nearing menopause, also called perimenopause.
- Hysterosonography. Hysterosonography (his-tur-o-suh-NOG-ruh-fee) uses sterile salt water called saline to expand the space inside the uterus, called the uterine cavity. This makes it easier to get images of submucosal fibroids and the lining of the uterus if you're trying to get pregnant or if you have heavy menstrual bleeding. Another name for hysterosonography is a saline infusion sonogram.
- Hysterosalpingography. Hysterosalpingography (his-tur-o-sal-ping-GOG-ruh-fee) uses a dye to highlight the uterine cavity and fallopian tubes on X-ray images. Your doctor may recommend it if infertility is a concern. This test can help find out if your fallopian tubes are open or are blocked, and it can show some submucosal fibroids.
- Hysteroscopy. For this exam, your doctor inserts a small, lighted telescope called a hysteroscope through your cervix into your uterus. Then saline is injected into your uterus. This expands the uterine cavity and lets your doctor check the walls of your uterus and the openings of your fallopian tubes.
More Information
Treatment
There's no single best treatment for uterine fibroids. Many treatment choices exist. If you have symptoms, talk with your care team about ways to get relief.
Watchful waiting
Many people with uterine fibroids have no symptoms. Or they have mildly annoying symptoms that they can live with. If that's the case for you, watchful waiting could be the best option.
Fibroids aren't cancer. They rarely interfere with pregnancy. They often grow slowly — or not at all — and tend to shrink after menopause, when levels of reproductive hormones drop.
Medicines
Medicines for uterine fibroids target hormones that control the menstrual cycle. They treat symptoms such as heavy menstrual bleeding and pelvic pressure. They don't get rid of fibroids, but they may shrink them. Medicines include:
-
Gonadotropin-releasing hormone (GnRH) agonists. These treat fibroids by blocking the body from making the hormones estrogen and progesterone. This puts you into a temporary menopause-like state. As a result, menstrual periods stop, fibroids shrink and anemia often gets better.
GnRH agonists include leuprolide (Lupron Depot, Eligard, others), goserelin (Zoladex) and triptorelin (Trelstar, Triptodur Kit).
Many people have hot flashes while using GnRH agonists. Often, these medicines are used for no more than six months. That's because symptoms return when the medicine is stopped, and long-term use can cause loss of bone. Sometimes, GnRH agonists are taken with low-dose estrogen or progestin. You might hear this called add-back therapy. It can ease side effects, and it might allow you to take GnRH agonists up to 12 months.
Your doctor may prescribe a GnRH agonist to shrink the size of your fibroids before a planned surgery. Or you might be prescribed this medicine to help transition you into menopause.
-
Gonadotropin-releasing hormone (GnRH) antagonists. These medicines can treat heavy menstrual bleeding in people with uterine fibroids who haven't gone through menopause. But they don't shrink fibroids. GnRH antagonists can be used for up to two years. Taking them along with add-back therapy can lessen side effects such as hot flashes and bone loss. Sometimes, low-dose estrogen or progestin are already included in these medicines.
GnRH antagonists include elagolix (Oriahnn) and relugolix (Myfembree).
- Progestin-releasing intrauterine device (IUD). A progestin-releasing IUD can relieve heavy bleeding caused by fibroids. It only relieves symptoms, though. It doesn't shrink fibroids or make them go away. It also prevents pregnancy.
- Tranexamic acid (Lysteda, Cyklokapron). This nonhormonal medicine can ease heavy menstrual periods. You take it only on heavy bleeding days.
-
Other medicines. Your doctor might recommend other medicines. For example, low-dose birth control pills can help control menstrual bleeding. But they don't reduce fibroid size.
Medicines called nonsteroidal anti-inflammatory drugs (NSAIDs) may help relieve pain tied to fibroids, but they don't reduce bleeding caused by fibroids. NSAIDs aren't hormonal medicines. Examples include ibuprofen (Advil, Motrin IB, others) and naproxen sodium (Aleve). Your doctor also may suggest that you take vitamins and iron if you have heavy menstrual bleeding and anemia.
Noninvasive procedure
Focused ultrasound surgery
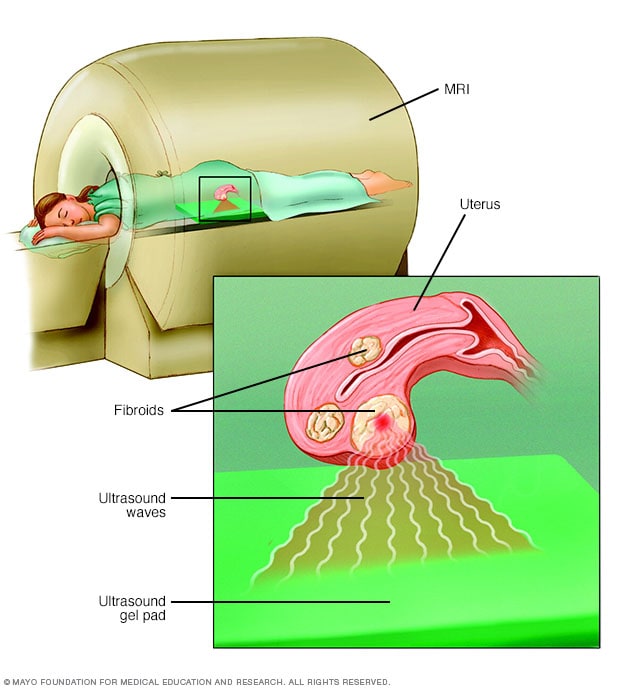
Focused ultrasound surgery
During focused ultrasound surgery, high-frequency, high-energy sound waves are used to target and destroy uterine fibroids. The procedure is performed while you're inside an MRI scanner. The equipment allows your doctor to visualize your uterus, locate any fibroids and destroy the fibroid tissue without making any incisions.
A noninvasive treatment doesn't involve surgical cuts called incisions. It also doesn't involve tools being placed in the body. With uterine fibroids, a procedure called MRI-guided focused ultrasound surgery (FUS) is:
- A noninvasive treatment option that preserves the uterus. It's done on an outpatient basis, meaning you don't have to spend the night at the hospital afterward.
- Done while you're inside an MRI scanner equipped with a high-energy ultrasound device for treatment. The images give your doctor the precise location of the uterine fibroids. When the location of the fibroid is targeted, the ultrasound device focuses sound waves into the fibroid to heat and destroy small areas of fibroid tissue.
- Newer technology, so researchers are learning more about the long-term safety and effectiveness. But so far data collected show that FUS for uterine fibroids is safe and works well. Still, it might not improve symptoms as much as a slightly more invasive procedure called uterine artery embolization could.
Minimally invasive procedures
Uterine artery embolization
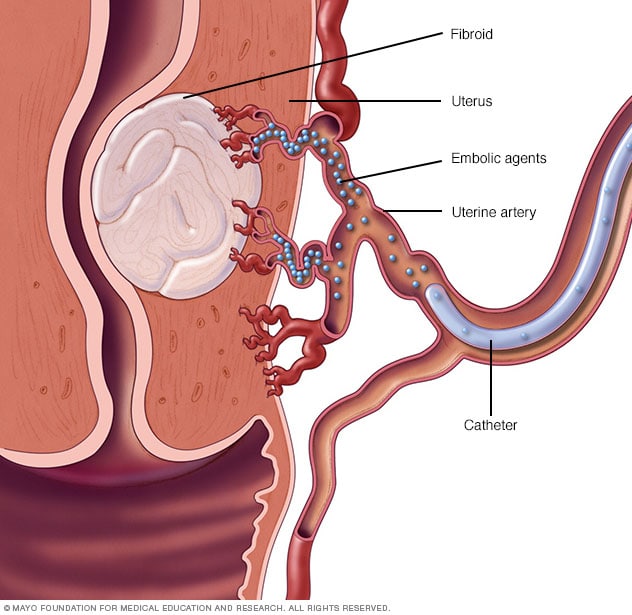
Uterine artery embolization
Small particles called embolic agents are injected into the uterine artery through a small catheter. The embolic agents then flow to the fibroids and lodge in the arteries that feed them. This cuts off blood flow to starve the tumors.
Laparoscopic radiofrequency ablation
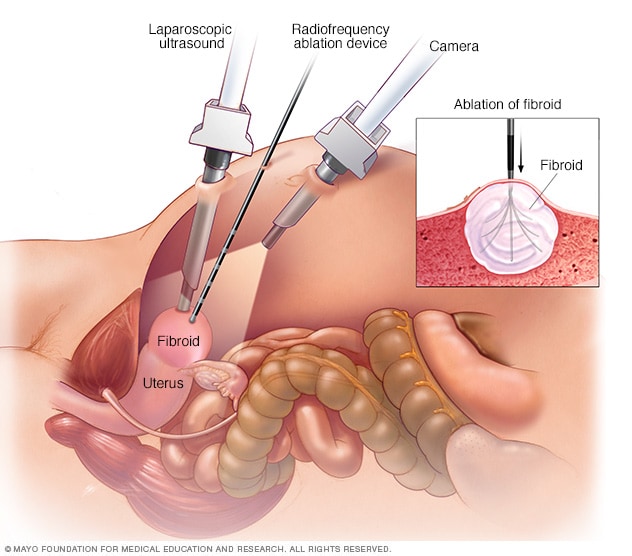
Laparoscopic radiofrequency ablation
During laparoscopic radiofrequency ablation, the doctor sees inside the abdomen using two special instruments. One is a laparoscopic camera positioned above the uterus. The other is a laparoscopic ultrasound wand that sits directly on the uterus. Using both instruments provides the doctor with two views of a uterine fibroid. This allows for more-thorough treatment than would be possible with just one view. After locating a uterine fibroid, the doctor uses another thin device to send several small needles into the fibroid. The small needles heat up, destroying fibroid tissue.
These procedures use no cuts or small cuts. They're linked with faster recovery times and fewer complications compared with traditional open surgery. Minimally invasive treatments for uterine fibroids include:
-
Uterine artery embolization. Small particles called embolic agents are injected into the arteries that supply the uterus with blood. The particles cut off blood flow to fibroids, causing them to shrink and die.
This technique can help shrink fibroids and relieve the symptoms they cause. Complications may happen if the blood supply to your ovaries or other organs is reduced. But research shows that complications are similar to surgical fibroid treatments. And the risk of needing a blood transfusion is lower.
-
Radiofrequency ablation. In this procedure, heat from radiofrequency energy destroys uterine fibroids and shrinks the blood vessels that feed them. This can be done through small cuts in the stomach area, a type of surgery called laparoscopy. It also can be done through the vagina, called a transvaginal procedure, or through the cervix, called a transcervical procedure.
With laparoscopic radiofrequency ablation, your doctor makes two small cuts in the abdomen. A slim viewing tool with a camera at the tip, called a laparoscope, is placed through the cuts. Using the camera and an ultrasound tool, your doctor finds fibroids to be treated.
After finding a fibroid, your doctor uses a device to send small needles into the fibroid. The needles heat up the fibroid tissue and destroy it. The destroyed fibroid changes right away. For instance, it goes from being hard like a golf ball to being soft like a marshmallow. During the next 3 to 12 months, the fibroid continues to shrink, and symptoms get better.
Laparoscopic radiofrequency ablation is also known as the Acessa procedure or Lap-RFA. Because there's no cutting of uterine tissue, doctors consider Lap-RFA a less invasive treatment than surgeries such as a hysterectomy and a myomectomy. Most people who have the procedure get back to regular activities within a few days.
The transcervical — or through the cervix — approach to radiofrequency ablation is called Sonata. It also uses ultrasound guidance to locate fibroids.
-
Laparoscopic or robotic myomectomy. In a myomectomy, your surgeon removes the fibroids and leaves the uterus in place.
If the fibroids are few in number, you and your doctor may opt for a laparoscopic procedure. This uses slender instruments placed through small cuts in the abdomen to remove the fibroids from the uterus.
Sometimes, a robotic system is used for the laparoscopic procedure. Your doctor views your stomach area on a monitor using a small camera attached to one of the instruments. Robotic myomectomy gives your surgeon a magnified, 3D view of your uterus. This can make the procedure more precise than is possible using some other techniques.
Larger fibroids can be removed through smaller cuts by breaking them into pieces with a device that cuts tissue. This is called morcellation. It can be done inside a surgical bag to lower the risk of spreading any cancer cells that doctors hadn't expected to find. Or it can be done by extending one incision to remove the fibroids without morcellation.
- Hysteroscopic myomectomy. This procedure may be an option if the fibroids are inside the uterus, also called submucosal fibroids. The fibroids are removed using tools placed through the vagina and cervix into the uterus.
-
Endometrial ablation. This procedure can reduce heavy menstrual flow. A device that's inserted into the uterus gives off heat, microwave energy, hot water, cold temperature or an electric current. This destroys the tissue that lines the inside of the uterus.
You aren't likely to get pregnant after endometrial ablation. But it's a good idea to take birth control to prevent a fertilized egg from forming in a fallopian tube, called an ectopic pregnancy. Without treatment, the growing tissue might cause life-threatening bleeding.
With any procedure that doesn't remove the uterus, there's a risk that new fibroids could grow and cause symptoms.
Traditional surgical procedures
Options for traditional open surgeries that use a larger incision include:
-
Abdominal myomectomy. This type of surgery removes fibroids through a larger cut in the stomach area, also called the abdomen. Your doctor may recommend it if you have more than one fibroid, very large fibroids or very deep fibroids.
Many people who are told that hysterectomy is their only option can have an abdominal myomectomy instead. Scarring after surgery can lower the chances of being able to get pregnant in the future, though.
-
Hysterectomy. This surgery removes the uterus. It remains the only proven permanent solution for uterine fibroids.
Hysterectomy ends your ability to bear children. If you also decide to have your ovaries removed, the surgery brings on menopause. You'll then choose whether to take hormone replacement therapy, which is medicine that can ease menopause side effects such as hot flashes. Most people with uterine fibroids may be able to choose to keep their ovaries.
Morcellation during fibroid removal
Morcellation is a process of breaking fibroids into smaller pieces. It can raise the risk of spreading cancer if a cancerous tumor that hadn't been found earlier gets broken up with morcellation during a myomectomy procedure. The risk can be lowered if:
- The surgical team looks into a person's risk factors before surgery.
- The fibroid is broken up in a surgical bag during morcellation.
- The incision is expanded to remove a large fibroid without morcellation.
All myomectomies carry the risk of cutting into cancer that hasn't been found. But younger people who haven't reached menopause by and large have a lower risk of undiagnosed cancer than do people over the age of 50.
Also, complications during open surgery are more common than the chance of spreading an unsuspected cancer in a fibroid during a minimally invasive procedure. If your doctor is planning to use morcellation, ask the doctor to explain your risks before treatment.
In the United States, the Food and Drug Administration (FDA) advises against the use of a morcellator device for most people who have fibroids removed through myomectomy or hysterectomy. The FDA recommends that people who are nearing menopause or who have reached menopause stay away from power morcellation. Older people who are in or entering menopause may have a higher cancer risk. And people who no longer want to get pregnant have other treatment options for fibroids.
If you're trying to get pregnant or might want to have children
With hysterectomy or endometrial ablation, you won't be able to get pregnant in the future. Also, uterine artery embolization and radiofrequency ablation may not be the best options if you want to keep as much of your fertility as possible.
Talk with your doctor about the risks and benefits of these procedures if you want to keep the ability to become pregnant. And if you're actively trying to get pregnant, get a complete fertility evaluation before you decide on a treatment plan for fibroids.
If fibroid treatment is needed — and you want to preserve your fertility — myomectomy is often the treatment of choice. But all treatments have risks and benefits. Talk about these with your doctor.
Risk of new fibroids
For all procedures except hysterectomy, seedlings — tiny tumors that your doctor doesn't detect during surgery — could one day grow and cause symptoms that need treatment. Often, this is called the recurrence rate. New fibroids also can form, and these may need treatment.
Also, some procedures may only treat some of the fibroids present at the time of treatment. These include laparoscopic or robotic myomectomy, radiofrequency ablation, and MRI-guided focused ultrasound surgery (FUS).
More Information
Clinical trials
Explore Mayo Clinic studies testing new treatments, interventions and tests as a means to prevent, detect, treat or manage this condition.
Alternative medicine
Some websites and health books promote alternative treatments for uterine fibroids. These include specific diet recommendations, magnet therapy, black cohosh, herbal preparations or homeopathy. So far, there's no scientific evidence to show that any of these techniques help.
Small studies suggest that acupuncture may help when used along with your main treatment for uterine fibroids. With this technique, a practitioner places very thin needles into certain points on the body.
Preparing for your appointment
Your first appointment likely will be with either your primary care doctor or a gynecologist. Appointments can be brief, so it's a good idea to prepare for your visit.
What you can do
- Make a list of any symptoms you have. Include all of your symptoms, even if you don't think they're related to the reason for your appointment.
- List any medicines, herbs and vitamin supplements you take. Include the amounts you take, called the doses, and how often you take them.
- Have a family member or close friend join you, if possible. You may be given a lot of information during your visit, and it can be tough to remember everything.
- Take a notebook or electronic device with you. Use it to note key information during your visit.
- Prepare a list of questions to ask. List your most important questions first, to be sure that you cover those points.
For uterine fibroids, some basic questions to ask include:
- How many fibroids do I have? How big are they and where are they located?
- What medicines are available to treat uterine fibroids or my symptoms?
- What side effects can I expect from medicine use?
- Under what circumstances do you recommend surgery?
- Will I need to take medicine before or after surgery?
- Will my uterine fibroids affect my ability to become pregnant?
- Can treatment of uterine fibroids improve my fertility?
Make sure that you understand everything your doctor tells you. Don't hesitate to have your doctor repeat information or to ask follow-up questions.
What to expect from your doctor
Some questions your doctor might ask include:
- How often do you have these symptoms?
- How long have you had them?
- How painful are your symptoms?
- Do your symptoms seem to be related to your menstrual cycle?
- Does anything make your symptoms better?
- Does anything make your symptoms worse?
- Do you have a family history of uterine fibroids?
Sept. 15, 2023