Diagnosis
Female cystoscopy
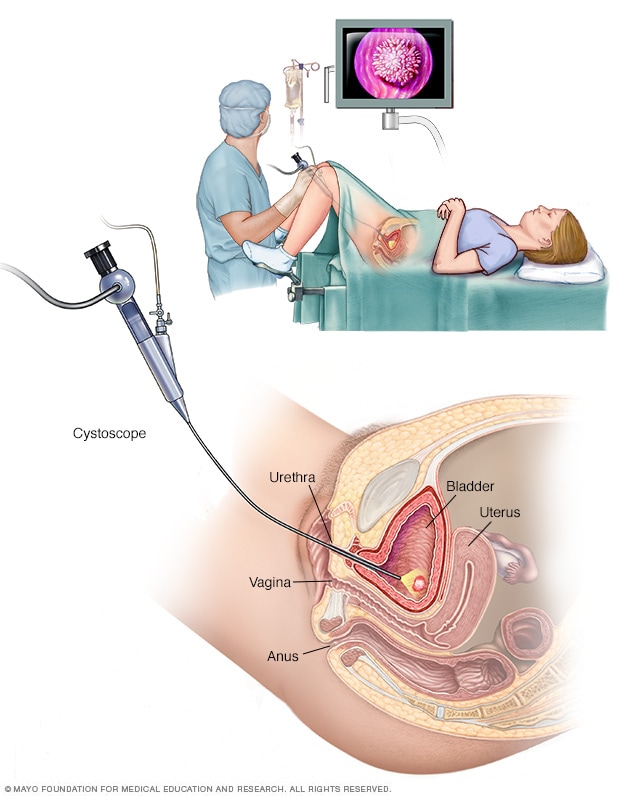
Female cystoscopy
Cystoscopy allows a health care provider to view the lower urinary tract to look for problems, such as a bladder stone. Surgical tools can be passed through the cystoscope to treat certain urinary tract conditions.
Male cystoscopy
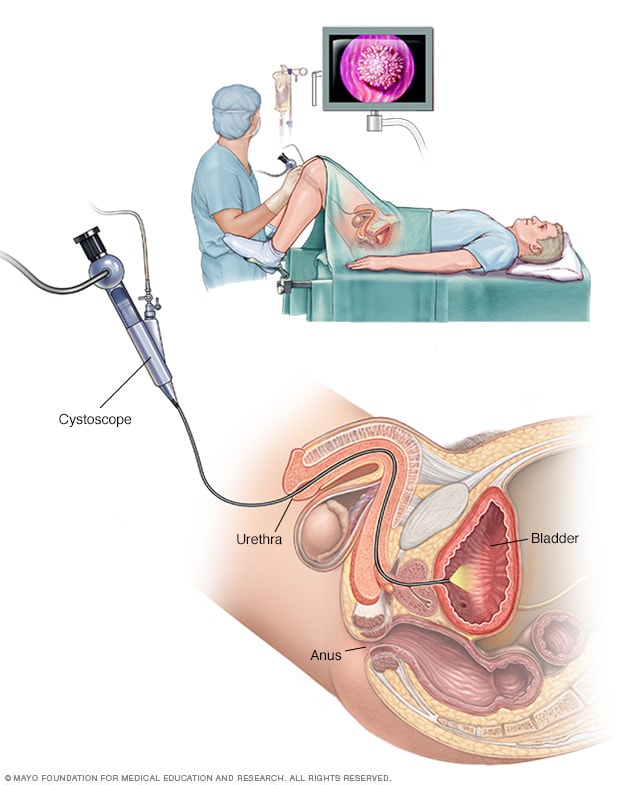
Male cystoscopy
Cystoscopy allows a health care provider to view the lower urinary tract to look for problems in the urethra and bladder. Surgical tools can be passed through the cystoscope to treat certain urinary tract conditions.
Tests and procedures used to diagnose urinary tract infections include:
- Analyzing a urine sample. Your health care provider may ask for a urine sample. The urine will be looked at in a lab to check for white blood cells, red blood cells or bacteria. You may be told to first wipe your genital area with an antiseptic pad and to collect the urine midstream. The process helps prevent the sample from being contaminated.
- Growing urinary tract bacteria in a lab. Lab analysis of the urine is sometimes followed by a urine culture. This test tells your provider what bacteria are causing the infection. It can let your provider know which medications will be most effective.
- Creating images of the urinary tract. Recurrent UTIs may be caused by a structural problem in the urinary tract. Your health care provider may order an ultrasound, a CT scan or MRI to look for this issue. A contrast dye may be used to highlight structures in your urinary tract.
- Using a scope to see inside the bladder. If you have recurrent UTIs, your health care provider may perform a cystoscopy. The test involves using a long, thin tube with a lens, called a cystoscope, to see inside the urethra and bladder. The cystoscope is inserted in the urethra and passed through to the bladder.
Treatment
Antibiotics usually are the first treatment for urinary tract infections. Your health and the type of bacteria found in your urine determine which medicine is used and how long you need to take it.
Simple infection
Medicines commonly used for simple UTIs include:
- Trimethoprim and sulfamethoxazole (Bactrim, Bactrim DS)
- Fosfomycin (Monurol)
- Nitrofurantoin (Macrodantin, Macrobid, Furadantin)
- Cephalexin
- Ceftriaxone
The group of antibiotics known as fluoroquinolones isn't commonly recommended for simple UTIs. These drugs include ciprofloxacin (Cipro), levofloxacin and others. The risks of these drugs generally outweigh the benefits for treating uncomplicated UTIs.
In cases of a complicated UTI or kidney infection, your health care provider might prescribe a fluoroquinolone medicine if there are no other treatment options.
Often, UTI symptoms clear up within a few days of starting treatment. But you may need to continue antibiotics for a week or more. Take all of the medicine as prescribed.
For an uncomplicated UTI that occurs when you're otherwise healthy, your health care provider may recommend a shorter course of treatment. That may mean taking an antibiotic for 1 to 3 days. Whether a short course of treatment is enough to treat your infection depends on your symptoms and medical history.
Your health care provider also may give you a pain reliever to take that can ease burning while urinating. But pain usually goes away soon after starting an antibiotic.
Frequent infections
If you have frequent UTIs, your health care provider may recommend:
- Low-dose antibiotics. You might take them for six months or longer.
- Diagnosing and treating yourself when symptoms occur. You'll also be asked to stay in touch with your provider.
- Taking a single dose of antibiotic after sex if UTIs are related to sexual activity.
- Vaginal estrogen therapy if you've reached menopause.
Severe infection
For a severe UTI, you may need IV antibiotics in a hospital.
Clinical trials
Explore Mayo Clinic studies testing new treatments, interventions and tests as a means to prevent, detect, treat or manage this condition.
Lifestyle and home remedies
Urinary tract infections can be painful, but you can take steps to ease discomfort until antibiotics treat the infection. Follow these tips:
- Drink plenty of water. Water helps to dilute your urine and flush out bacteria.
- Avoid drinks that may irritate your bladder. Avoid coffee, alcohol, and soft drinks containing citrus juices or caffeine until the infection has cleared. They can irritate your bladder and tend to increase the need to urinate.
- Use a heating pad. Apply a warm, but not hot, heating pad to your belly to help with bladder pressure or discomfort.
Alternative medicine
Many people drink cranberry juice to prevent UTIs. There's some indication that cranberry products, in either juice or tablet form, may have properties that fight an infection. Researchers continue to study the ability of cranberry juice to prevent UTIs, but results aren't final.
There's little harm in drinking cranberry juice if you feel it helps you prevent UTIs, but watch the calories. For most people, drinking cranberry juice is safe. However, some people report an upset stomach or diarrhea.
But don't drink cranberry juice if you're taking blood-thinning medication, such as warfarin (Jantovin).
Preparing for your appointment
Your primary care provider, nurse practitioner or other health care provider can treat most UTIs. If you have frequent UTIs or a chronic kidney infection, you may be referred to a health care provider who specializes in urinary disorders. This type of doctor is called a urologist. Or you may see a health care provider who specializes in kidney disorders. This type of doctor is called a nephrologist.
What you can do
To get ready for your appointment:
- Ask if there's anything you need to do in advance, such as collect a urine sample.
- Take note of your symptoms, even if you're not sure they're related to a UTI.
- Make a list of all the medicines, vitamins or other supplements that you take.
- Write down questions to ask your health care provider.
For a UTI, basic questions to ask your provider include:
- What's the most likely cause of my symptoms?
- Are there any other possible causes?
- Do I need any tests to confirm the diagnosis?
- What factors do you think may have contributed to my UTI?
- What treatment approach do you recommend?
- If the first treatment doesn't work, what will you recommend next?
- Am I at risk of complications from this condition?
- What is the risk that this problem will come back?
- What steps can I take to lower the risk of the infection coming back?
- Should I see a specialist?
Don't hesitate to ask other questions as they occur to you during your appointment.
What to expect from your doctor
Your health care provider will likely ask you several questions, including:
- When did you first notice your symptoms?
- Have you ever been treated for a bladder or kidney infection?
- How severe is your discomfort?
- How often do you urinate?
- Are your symptoms relieved by urinating?
- Do you have low back pain?
- Have you had a fever?
- Have you noticed vaginal discharge or blood in your urine?
- Are you sexually active?
- Do you use contraception? What kind?
- Could you be pregnant?
- Are you being treated for any other medical conditions?
- Have you ever used a catheter?
Sept. 14, 2022