Diagnosis
Get answers to the most frequently asked questions about prostate cancer from Mayo Clinic urologist Mitchell Humphreys, M.D.
Hello. I'm Dr. Humphreys, a urologist at Mayo Clinic, and I'm here to answer some of the important questions you may have about prostate cancer.
How do you know how fast my cancer is growing?
If you have low to intermediate risk prostate cancer, there are genomic tests that can better inform on the risk of developing a more aggressive cancer. These tests look at the DNA of your actual cancer cells to compare them to other men, to come up with an individual risk profile for you and your cancer. None of it is 100%, but it does provide the best evidence based on your specific prostate cancer.
Is prostate cancer sexually transmitted?
No, there's no risk to your partner from prostate cancer. There's no risk with sexual activity. Prostate cancer is internal and does not spread through contact.
Is prostate cancer hereditary?
Some prostate cancers are hereditary. If you have prostate cancer, all of your first-degree relatives -- parent, sibling, or child -- are at an elevated risk for developing prostate cancer. If you are diagnosed when you're young in your 40s and develop prostate cancer, you may want to consider a genetic consultation to see if there are any known genetic risk factors that you and your family may have.
What can I do to prevent or slow prostate cancer?
There's no one thing. A healthy lifestyle with 30 minutes of exercise a day has shown to be protective. Also, diet is important by limiting red meat and eating fresh fruits and vegetables, low in sugars and carbohydrates. I would advise following a heart-healthy diet as research has shown that it is healthy for the prostate as well.
Is there a risk of cancer spreading if I have a biopsy of my prostate?
No, prostate cancer doesn't spread that way. And there have been millions of biopsies throughout the world and never a single incident of it being spread that way has ever been reported.
When should I stop screening for prostate cancer?
Not all prostate cancer is lethal and not all prostate cancer requires treatment. As a general rule of thumb, if your life expectancy is 10 years or less, you probably will not have to worry about prostate cancer affecting you in your lifetime. However, you should discuss this with your care team to determine how it specifically relates to you.
How can I be the best partner to my medical team?
The best thing you can do is be open and honest. Your medical team is here to be a resource to you, to support you and to help you in any way they can. Never hesitate to ask your medical team any questions or concerns that you have being informed makes all the difference. Thank you for your time and we wish you well.
Screening for prostate cancer
Digital rectal exam
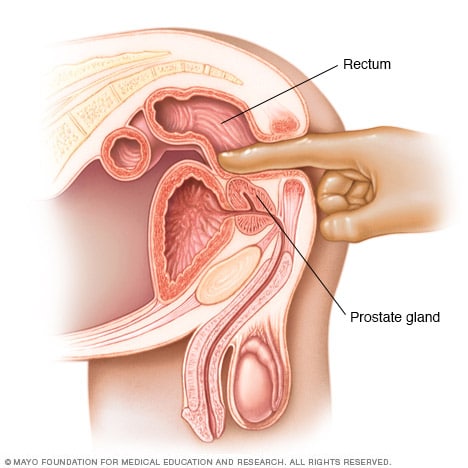
Digital rectal exam
During a digital rectal exam, your doctor inserts a gloved, lubricated finger into your rectum and feels the back wall of the prostate gland for enlargement, tenderness, lumps or hard spots.
Testing healthy men with no symptoms for prostate cancer is controversial. There is some disagreement among medical organizations whether the benefits of testing outweigh the potential risks.
Most medical organizations encourage men in their 50s to discuss the pros and cons of prostate cancer screening with their doctors. The discussion should include a review of your risk factors and your preferences about screening.
You might consider starting the discussions sooner if you're a Black person, have a family history of prostate cancer or have other risk factors.
Prostate screening tests might include:
- Digital rectal exam (DRE). During a DRE, your doctor inserts a gloved, lubricated finger into your rectum to examine your prostate, which is adjacent to the rectum. If your doctor finds any abnormalities in the texture, shape or size of the gland, you may need further tests.
- Prostate-specific antigen (PSA) test. A blood sample is drawn from a vein in your arm and analyzed for PSA, a substance that's naturally produced by your prostate gland. It's normal for a small amount of PSA to be in your bloodstream. However, if a higher than usual level is found, it may indicate prostate infection, inflammation, enlargement or cancer.
Diagnosing prostate cancer
Transrectal biopsy of the prostate
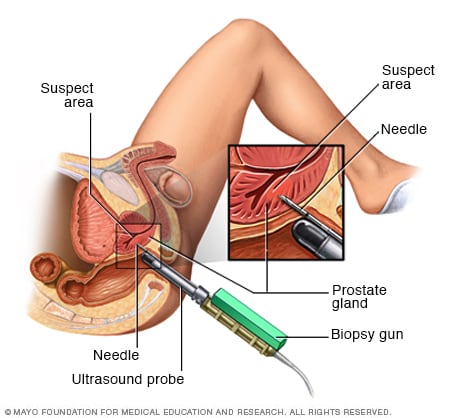
Transrectal biopsy of the prostate
During a transrectal biopsy, a biopsy gun quickly projects a thin needle into suspect areas of the prostate gland, and small sections of tissue are removed for analysis.
If prostate cancer screening detects an abnormality, your doctor may recommend further tests to determine whether you have prostate cancer, such as:
- Ultrasound. During a transrectal ultrasound, a small probe, about the size and shape of a cigar, is inserted into your rectum. The probe uses sound waves to create a picture of your prostate gland.
- Magnetic resonance imaging (MRI). In some situations, your doctor may recommend an MRI scan of the prostate to create a more detailed picture. MRI images may help your doctor plan a procedure to remove prostate tissue samples.
- Collecting a sample of prostate tissue. To determine whether there are cancer cells in the prostate, your doctor may recommend a procedure to collect a sample of cells from your prostate (prostate biopsy). Prostate biopsy is often done using a thin needle that's inserted into the prostate to collect tissue. The tissue sample is analyzed in a lab to determine whether cancer cells are present.
Determining whether prostate cancer is aggressive
When a biopsy confirms the presence of cancer, the next step is to determine the level of aggressiveness (grade) of the cancer cells. A doctor in a lab examines a sample of your cancer cells to determine how much cancer cells differ from the healthy cells. A higher grade indicates a more aggressive cancer that is more likely to spread quickly.
Techniques used to determine the aggressiveness of the cancer include:
-
Gleason score. The most common scale used to evaluate the grade of prostate cancer cells is called a Gleason score. Gleason scoring combines two numbers and can range from 2 (nonaggressive cancer) to 10 (very aggressive cancer), though the lower part of the range isn't used as often.
Most Gleason scores used to assess prostate biopsy samples range from 6 to 10. A score of 6 indicates a low-grade prostate cancer. A score of 7 indicates a medium-grade prostate cancer. Scores from 8 to 10 indicate high-grade cancers.
- Genomic testing. Genomic testing analyzes your prostate cancer cells to determine which gene mutations are present. This type of test can give you more information about your prognosis. But it's not clear who might benefit most from this information, so the tests aren't widely used. Genomic tests aren't necessary for every person with prostate cancer, but they might provide more information for making treatment decisions in certain situations.
Determining whether the cancer has spread
Once a prostate cancer diagnosis has been made, your doctor works to determine the extent (stage) of the cancer. If your doctor suspects your cancer may have spread beyond your prostate, one or more of the following imaging tests may be recommended:
- Bone scan.
- Ultrasound.
- Computerized tomography (CT) scan.
- Magnetic resonance imaging (MRI).
- Positron emission tomography (PET) scan.
Not every person should have every test. Your doctor will help determine which tests are best for your individual situation.
Your doctor uses the information from these tests to assign your cancer a stage. Prostate cancer stages are indicated by Roman numerals ranging from I to IV. The lowest stages indicate the cancer is confined to the prostate. By stage IV, the cancer has grown beyond the prostate and may have spread to other areas of the body.
More Information
Treatment
Your prostate cancer treatment options depend on several factors, such as how fast your cancer is growing, whether it has spread and your overall health, as well as the potential benefits or side effects of the treatment.
Immediate treatment may not be necessary
Low-grade prostate cancer may not need treatment right away. For some, treatment may never be needed. Instead, doctors sometimes recommend active surveillance.
In active surveillance, regular follow-up blood tests, rectal exams and prostate biopsies may be performed to monitor progression of your cancer. If tests show your cancer is progressing, you may opt for a prostate cancer treatment such as surgery or radiation.
Active surveillance may be an option for cancer that isn't causing symptoms, is expected to grow very slowly and is confined to a small area of the prostate. Active surveillance may also be considered for someone who has another serious health condition or who is of an advanced age that makes cancer treatment more difficult.
Surgery to remove the prostate
Prostatectomy incisions
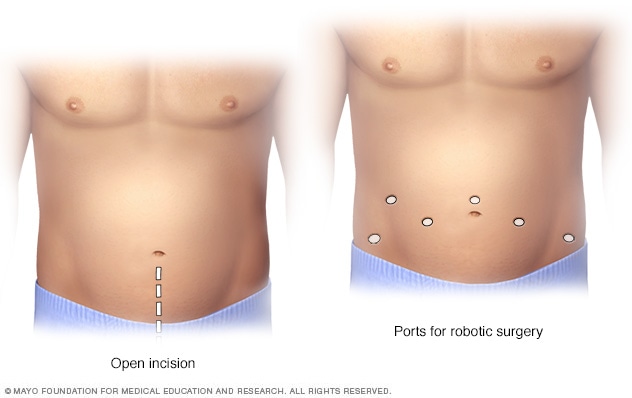
Prostatectomy incisions
During an open prostatectomy, one large incision is made in the lower stomach area (left). During a robotic prostatectomy, several smaller incisions are made in the stomach area (right).
Surgery for prostate cancer involves removing the prostate gland (radical prostatectomy), some surrounding tissue and a few lymph nodes.
Surgery is an option for treating cancer that's confined to the prostate. It's sometimes used to treat advanced prostate cancer in combination with other treatments.
To access the prostate, surgeons may use a technique that involves:
- Making several small incisions in your abdomen. During robot-assisted laparoscopic prostatectomy, surgical instruments are attached to a mechanical device (robot) and inserted through several small incisions in your abdomen. The surgeon sits at a console and uses hand controls to guide the robot to move the instruments. Most prostate cancer operations are done using this technique.
- Making one long incision in your abdomen. During retropubic surgery, the surgeon makes one long incision in your lower abdomen to access and remove the prostate gland. This approach is much less common, but may be necessary in certain situations.
Discuss with your doctor which type of surgery is best for your specific situation.
Radiation therapy
External beam radiation for prostate cancer
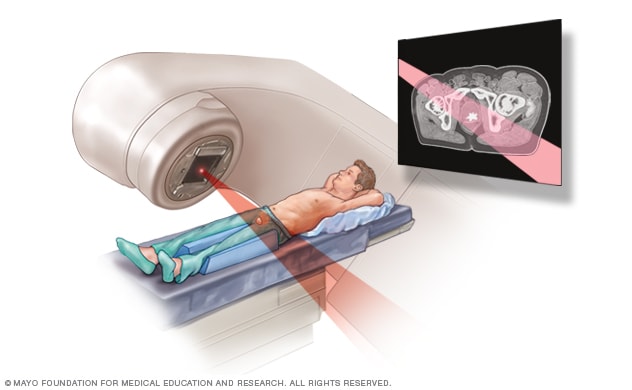
External beam radiation for prostate cancer
During external beam radiation treatment for prostate cancer, you lie on a table while a linear accelerator moves around you to deliver radiation from many angles. The linear accelerator delivers the precise dose of radiation planned by your treatment team.
Permanent prostate brachytherapy
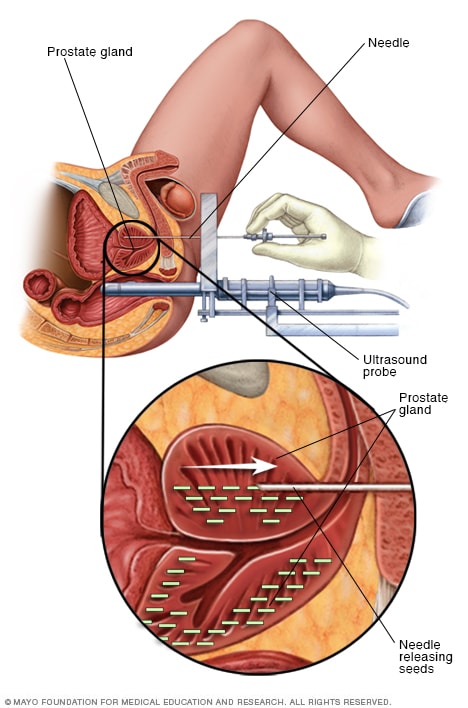
Permanent prostate brachytherapy
Permanent prostate brachytherapy involves placing many radioactive seeds within the prostate to treat prostate cancer. During the procedure, an ultrasound probe is placed in the rectum to help guide the placement of seeds. The seeds emit radiation that dissipates over a few months.
Radiation therapy uses high-powered energy to kill cancer cells. Prostate cancer radiation therapy treatments may involve:
-
Radiation that comes from outside of your body (external beam radiation). During external beam radiation therapy, you lie on a table while a machine moves around your body, directing high-powered energy beams, such as X-rays or protons, to your prostate cancer. You typically undergo external beam radiation treatments five days a week for several weeks. Some medical centers offer a shorter course of radiation therapy that uses higher doses of radiation spread over fewer days.
External beam radiation is an option for treating cancer that's confined to the prostate. It can also be used after surgery to kill any cancer cells that might remain if there's a risk that the cancer could spread or come back. For prostate cancer that spreads to other areas of the body, such as the bones, radiation therapy can help slow the cancer's growth and relieve symptoms, such as pain.
- Radiation placed inside your body (brachytherapy). Brachytherapy involves placing radioactive sources in your prostate tissue. Most often, the radiation is contained in rice-sized radioactive seeds that are inserted into your prostate tissue. The seeds deliver a low dose of radiation over a long period of time. Brachytherapy is one option for treating cancer that hasn't spread beyond the prostate.
In some situations, doctors may recommend both types of radiation therapy.
Freezing or heating prostate tissue
Ablative therapies destroy prostate tissue with cold or heat. Options may include:
- Freezing prostate tissue. Cryoablation or cryotherapy for prostate cancer involves using a very cold gas to freeze the prostate tissue. The tissue is allowed to thaw and the procedure repeats. The cycles of freezing and thawing kill the cancer cells and some surrounding healthy tissue.
- Heating prostate tissue. High-intensity focused ultrasound (HIFU) treatment uses concentrated ultrasound energy to heat the prostate tissue and cause it to die.
These treatments may be considered for treating very small prostate cancers when surgery isn't possible. They may also be used to treat advanced prostate cancers if other treatments, such as radiation therapy, haven't helped.
Researchers are studying whether cryotherapy or HIFU to treat one part of the prostate might be an option for cancer that's confined to the prostate. Referred to as "focal therapy," this strategy identifies the area of the prostate that contains the most aggressive cancer cells and treats that area only. Studies have found that focal therapy reduces the risk of side effects. But it's not clear whether it offers the same survival benefits as treatment to the entire prostate.
Hormone therapy
Hormone therapy is treatment to stop your body from producing the male hormone testosterone. Prostate cancer cells rely on testosterone to help them grow. Cutting off the supply of testosterone may cause cancer cells to die or to grow more slowly.
Hormone therapy options include:
- Medications that stop your body from producing testosterone. Certain medications — known as luteinizing hormone-releasing hormone (LHRH) or gonadotropin-releasing hormone (GnRH) agonists and antagonists — prevent your body's cells from receiving messages to make testosterone. As a result, your testicles stop producing testosterone.
- Medications that block testosterone from reaching cancer cells. These medications, known as anti-androgens, usually are given in conjunction with LHRH agonists. That's because LHRH agonists can cause a temporary increase in testosterone before testosterone levels decrease.
- Surgery to remove the testicles (orchiectomy). Removing your testicles reduces testosterone levels in your body quickly and significantly. But unlike medication options, surgery to remove the testicles is permanent and irreversible.
Hormone therapy is often used to treat advanced prostate cancer to shrink the cancer and slow its growth.
Hormone therapy is sometimes used before radiation therapy to treat cancer that hasn't spread beyond the prostate. It helps shrink the cancer and increases the effectiveness of radiation therapy.
Chemotherapy
Chemotherapy uses drugs to kill rapidly growing cells, including cancer cells. Chemotherapy can be administered through a vein in your arm, in pill form or both.
Chemotherapy may be a treatment option for treating prostate cancer that has spread to other areas of the body. Chemotherapy may also be an option for cancers that don't respond to hormone therapy.
Immunotherapy
Immunotherapy uses your immune system to fight cancer. Your body's disease-fighting immune system may not attack your cancer because the cancer cells produce proteins that help them hide from the immune system cells. Immunotherapy works by interfering with that process.
Prostate cancer immunotherapy can involve:
- Engineering your cells to fight cancer. Sipuleucel-T (Provenge) treatment takes some of your own immune cells, genetically engineers them in a laboratory to fight prostate cancer and then injects the cells back into your body through a vein. It's an option for treating advanced prostate cancer that no longer responds to hormone therapy.
- Helping your immune system cells identify cancer cells. Immunotherapy drugs that help the immune system cells identify and attack the cancer cells are an option for treating advanced prostate cancers that no longer respond to hormone therapy.
Targeted drug therapy
Targeted drug treatments focus on specific abnormalities present within cancer cells. By blocking these abnormalities, targeted drug treatments can cause cancer cells to die.
Targeted therapy drugs may be recommended to treat advanced or recurrent prostate cancer if hormone therapy isn't working.
Some targeted therapies only work in people whose cancer cells have certain genetic mutations. Your cancer cells may be tested in a laboratory to see if these drugs might help you.
More Information
Clinical trials
Explore Mayo Clinic studies testing new treatments, interventions and tests as a means to prevent, detect, treat or manage this condition.
Alternative medicine
No complementary or alternative treatments will cure prostate cancer. However, complementary and alternative prostate cancer treatments may help you cope with the side effects of cancer and its treatment.
Nearly everyone diagnosed with cancer experiences some distress at some point. If you're distressed, you may feel sad, angry or anxious. You may experience difficulty sleeping or find yourself constantly thinking about your cancer.
Several complementary medicine techniques may help you cope with your distress, including:
- Art therapy.
- Dance or movement therapy.
- Exercise.
- Meditation.
- Music therapy.
- Relaxation techniques.
- Spirituality.
Discuss your feelings and concerns with your doctor. In some cases, treatment for distress may require medications.
Coping and support
When you receive a diagnosis of prostate cancer, you may experience a range of feelings — including disbelief, fear, anger, anxiety and depression. With time, each person finds his own way of coping with a prostate cancer diagnosis.
Until you find what works for you, try to:
- Learn enough about prostate cancer to feel comfortable making treatment decisions. Learn as much as you need to know about your cancer and its treatment in order to understand what to expect from treatment and life after treatment. Ask your doctor, nurse or other health care professional to recommend some reliable sources of information to get you started.
- Keep your friends and family close. Your friends and family can provide support during and after your treatment. They may be eager to help with the small tasks you won't have energy for during treatment. And having a close friend or family member to talk to can be helpful when you're feeling stressed or overwhelmed.
- Connect with other cancer survivors. Friends and family can't always understand what it's like to face cancer. Other cancer survivors can provide a unique network of support. Ask your health care providers about support groups or community organizations that can connect you with other cancer survivors. Organizations such as the American Cancer Society offer online chat rooms and discussion forums.
- Take care of yourself. Take care of yourself during cancer treatment by eating a diet full of fruits and vegetables. Try to exercise most days of the week. Get enough sleep each night so that you wake feeling rested.
- Continue sexual expression. If you experience erectile dysfunction, your natural reaction may be to avoid all sexual contact. But consider touching, holding, hugging and caressing as ways to continue sharing sexuality with your partner.
Preparing for your appointment
If you have signs or symptoms that worry you, start by seeing your family doctor.
If your doctor suspects you may have a problem with your prostate, you may be referred to a urinary tract specialist (urologist). If you're diagnosed with prostate cancer, you may be referred to a cancer specialist (oncologist) or a specialist who uses radiation therapy to treat cancer (radiation oncologist).
Because appointments can be brief, and because there's often a lot of information to discuss, it's a good idea to be prepared. Here's some information to help you get ready and what to expect from your doctor.
What you can do
- Be aware of any pre-appointment restrictions. At the time you make the appointment, be sure to ask if there's anything you need to do in advance, such as restrict your diet.
- Write down any symptoms you're experiencing, including any that may seem unrelated to the reason for which you scheduled the appointment.
- Write down key personal information, including any major stresses or recent life changes.
- Make a list of all medications, vitamins or supplements that you're taking.
- Consider taking a family member or friend along. Sometimes it can be difficult to remember all the information provided during an appointment. Someone who accompanies you may remember something that you missed or forgot.
- Write down questions to ask your doctor.
Your time with your doctor is limited, so preparing a list of questions can help you make the most of your time together. List your questions from most important to least important in case time runs out. For prostate cancer, some basic questions to ask your doctor include:
- Do I have prostate cancer?
- How large is my prostate cancer?
- Has my prostate cancer spread beyond my prostate?
- What's my Gleason score?
- What's my prostate-specific antigen (PSA) level?
- Will I need more tests?
- What are my treatment options?
- Is there one treatment option you think is best for me?
- Do I need cancer treatment right away, or is it possible to wait and see if the cancer grows?
- What are the potential side effects of each treatment?
- What is the chance that my prostate cancer will be cured with treatment?
- If you had a friend or family member in my situation, what would you recommend?
- Should I see a specialist? What will that cost, and will my insurance cover it?
- Are there brochures or other printed material that I can take with me? What websites do you recommend?
In addition to the questions that you've prepared to ask your doctor, don't hesitate to ask other questions during your appointment.
What to expect from your doctor
Your doctor is likely to ask you a number of questions. Being ready to answer them may allow more time later to cover other points you want to address. Your doctor may ask:
- When did you first begin experiencing symptoms?
- Have your symptoms been continuous or occasional?
- How severe are your symptoms?
- What, if anything, seems to improve your symptoms?
- What, if anything, appears to worsen your symptoms?