Overview
Postherpetic neuralgia (post-hur-PET-ik noo-RAL-juh) is the most common complication of shingles. It causes a burning pain in nerves and skin. The pain lasts long after the rash and blisters of shingles go away.
The risk of postherpetic neuralgia rises with age. It mainly affects people older than 60. There's no cure, but treatments can ease symptoms. For most people, postherpetic neuralgia gets better over time.
Products & Services
Symptoms
In general, the symptoms of postherpetic neuralgia are limited to the area of skin where the shingles outbreak first happened. That's commonly in a band around the trunk of the body, most often on one side.
The symptoms might include:
- Pain that lasts three months or longer after the shingles rash has healed. The pain may feel burning, sharp and jabbing. Or it may feel deep and aching.
- Not being able to stand light touch. People with postherpetic neuralgia often can't bear even the touch of clothing on the affected skin.
- Itching or loss of feeling. Less often, postherpetic neuralgia can cause an itchy feeling or numbness.
When to see a doctor
See a health care provider at the first sign of shingles. Often the pain starts before you notice a rash. The risk of postherpetic neuralgia becomes lower if you start taking virus-fighting medicines called antivirals within 72 hours of getting the shingles rash.
Causes
Shingles affects the nerves
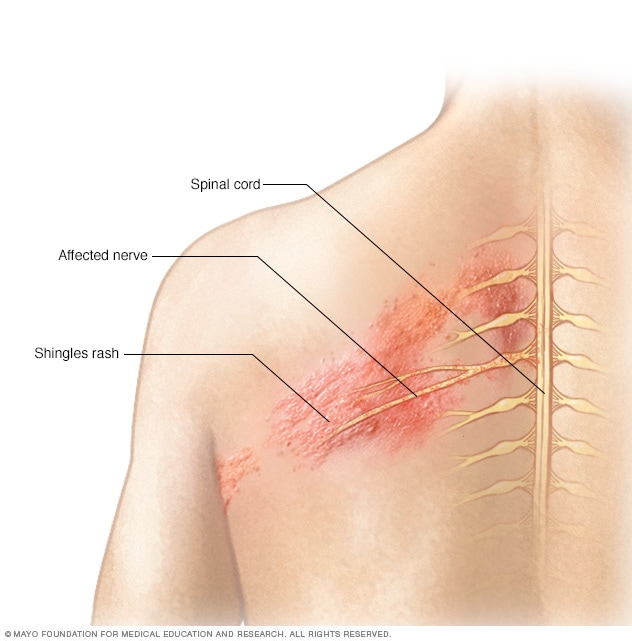
Shingles affects the nerves
The shingles rash is associated with an inflammation of nerves beneath the skin.
Illustration of shingles rash on different skin colors

Illustration of shingles rash on different skin colors
The shingles rash is associated with an inflammation of nerves beneath the skin.
The chickenpox virus causes shingles. Once you've had chickenpox, the virus stays in your body for the rest of your life. The virus can become active again and cause shingles. This risk goes up with age. The risk also rises if something turns down the body's immune system, such as chemotherapy medicines to treat cancer.
Postherpetic neuralgia happens if nerve fibers get damaged during an outbreak of shingles. Damaged fibers can't send messages from the skin to the brain as they usually do. Instead, the messages become confused and heightened. This causes pain that can last months or even years.
Risk factors
With shingles, things that can raise the risk of postherpetic neuralgia are:
- Age. You're older than 60.
- How bad the case of shingles was. You had a severe rash and pain that kept you from doing daily activities.
- Other illness. You have a long-term disease, such as diabetes.
- Where shingles appeared. You had shingles on your face or torso.
- A delay in shingles treatment. You didn't start taking antiviral medicine within 72 hours of your rash appearing.
- No shingles vaccine. You had not been vaccinated for shingles.
Complications
People with postherpetic neuralgia can develop other problems that are common with long-term pain. It depends on how long the postherpetic neuralgia lasts and how painful it is. These other problems can include:
- Depression.
- Trouble sleeping.
- Tiredness
- Not feeling as hungry as usual.
Prevention
Shingles vaccines can help prevent shingles and postherpetic neuralgia. Ask your health care provider when you should get a vaccine.
In the United States, the Centers for Disease Control and Prevention (CDC) suggests that adults 50 and older get a shingles vaccine called Shingrix. The agency also suggests Shingrix for adults 19 and older who have weaker immune systems because of diseases or treatments. Shingrix is suggested even if you've already had shingles or the older vaccine, Zostavax. Shingrix is given in two doses, 2 to 6 months apart.
With two doses, Shingrix is more than 90% effective in preventing shingles and postherpetic neuralgia. Other shingles vaccines are offered outside of the United States. Talk to your provider for more information on how well they prevent shingles and postherpetic neuralgia.
Feb. 10, 2023