Diagnosis
Because NAFLD typically causes no symptoms, it is often found when tests done for other reasons point to a liver problem. For example, a blood test done during a yearly exam may show high levels of liver enzymes, which can lead to more testing and a NAFLD diagnosis.
Tests done to diagnosis NAFLD, rule out other diseases and see how bad liver damage is include:
Blood tests
- Complete blood count.
- Iron studies, which show how much iron is in your blood and other cells.
- Liver enzyme and liver function tests.
- Tests for chronic viral hepatitis (hepatitis A, hepatitis C and others).
- Celiac disease screening test.
- Fasting blood sugar.
- Hemoglobin A1C, which shows how stable your blood sugar is.
- Lipid profile, which measures blood fats, such as cholesterol and triglycerides.
Imaging procedures
Imaging tests used to diagnose NAFLD include:
- Abdominal ultrasound, which is often the first test used when liver disease is suspected.
- Magnetic resonance imaging (MRI) or computerized tomography (CT) scanning. These tests are better at finding mild liver fibrosis but can't tell NASH from NAFLD.
- Transient elastography, a newer type of ultrasound that measures the stiffness of your liver. Liver stiffness is a sign of fibrosis or scarring.
- Magnetic resonance elastography, which combines MRI imaging with sound waves to create a visual map, or elastogram, showing the stiffness of body tissues.
Liver biopsy
If other tests show signs of more-advanced liver disease or NASH, or if your test results are unclear, your doctor may suggest a liver biopsy. Liver biopsy is a procedure to remove a small piece of tissue from your liver. It is usually done using a needle through the abdominal wall. The tissue sample is looked at in a lab for signs of inflammation and scarring. Liver biopsy is the best way to diagnose NASH and clearly shows the amount of liver damage.
A liver biopsy can be uncomfortable, and it does have risks that your health care team will go over with you in detail. This procedure is done using a needle that is passed through the abdominal wall and into the liver.
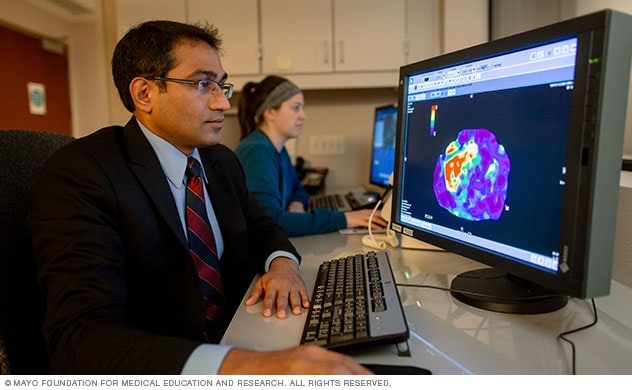
A Mayo Clinic radiologist views a magnetic resonance elastogram of the liver showing areas of scarring, or fibrosis, in red.
More Information
Treatment
Treatment for NAFLD usually starts with weight loss. This can be done by eating a healthy diet, limiting portion sizes and exercise. Losing weight may improve other health problems that lead to NAFLD. Typically, losing 10% of your body weight or more is recommended. But losing even 3% to 5% of your starting weight can have benefits. Weight-loss surgery or medicines also may be helpful for certain people.
A new medicine is available to treat people who have NASH with moderate to severe liver scarring. Resmetirom (Rezdiffra) can help reduce the amount of fat that collects in the liver. This medicine is not recommended for people with cirrhosis. For those who have cirrhosis due to NASH, a liver transplant may be needed.
Clinical trials
Explore Mayo Clinic studies testing new treatments, interventions and tests as a means to prevent, detect, treat or manage this condition.
Lifestyle and home remedies
With help from your health care team, you can take steps to manage nonalcoholic fatty liver disease. You can:
- Lose weight. If you're overweight or obese, reduce the number of calories you eat each day and increase your physical activity to lose weight slowly. Eating fewer calories is key to losing weight and managing this disease. If you tried to lose weight in the past and couldn't, ask your health care team for help.
- Choose a healthy diet. Eat a healthy diet that's rich in fruits, vegetables and whole grains. Your health care team may suggest avoiding or limiting certain foods and drinks, such as white bread, red and processed meats, juices, and sweetened drinks. Keep track of all calories you take in.
- Exercise and be more active. Aim for at least 150 minutes of exercise a week. If you're trying to lose weight, you might find that more exercise is helpful. But if you don't already exercise regularly, get your health care team's OK first and start slowly.
- Manage your diabetes. Follow your health care team's advice to manage your diabetes. Take your medicines as told by your care team and watch your blood sugar closely.
- Lower your cholesterol and blood pressure. Improve your cholesterol levels and blood pressure if they are high. A healthy diet, exercise and medicines can help keep your cholesterol, triglycerides and blood pressure at healthy levels.
- Protect your liver. Avoid things that could harm your liver health. For example, don't drink alcohol. Follow the instructions on all medicines and nonprescription drugs. Check with your health care team before using any herbal supplements, as some can harm the liver.
Alternative medicine
No alternative medicine treatments are proved to cure nonalcoholic fatty liver disease. But researchers are studying whether some supplements or natural compounds could be helpful, such as:
-
Vitamin E. In theory, vitamin E and other vitamins called antioxidants could help protect the liver by reducing or canceling out the damage caused by inflammation. But more research is needed.
Some evidence suggests vitamin E supplements may be helpful for people with NAFLD who don't have type 2 diabetes. Vitamin E supplements are not recommended for people with serious liver scarring or type 2 diabetes. Vitamin E has been linked with a slightly increased risk of heart disease and prostate cancer.
-
Caffeinated coffee. Some studies suggest that coffee may benefit the liver by reducing the risk of liver diseases like NAFLD and lowering the chance of scarring. It's not yet clear how coffee may prevent liver damage. But certain compounds in coffee are thought to lower inflammation and slow scar tissue growth.
If you already drink coffee, these results may make you feel better about your morning cup. But if you don't already drink coffee, this probably isn't a good reason to start. Discuss the possible benefits of coffee with your health care team.
Preparing for your appointment
See your family doctor or primary doctor first if you have symptoms that worry you. If your doctor suspects a liver problem, such as nonalcoholic fatty liver disease, you may be referred to a doctor who specializes in the liver, called a hepatologist.
Because appointments can be short, it's a good idea to be well prepared. Here are a few tips to help you get ready, and what to expect from your doctor.
What you can do
- Know what to do before your visit. When you make the appointment, ask if there's anything you need to do beforehand.
- Write down any symptoms you're having, including any that seem unrelated to the appointment.
- Make a list of all medications, vitamins or supplements you're taking.
- Take any relevant medical records, such as records of any tests you've had that relate to your current condition.
- Take a family member or friend along, if possible. Sometimes it can be hard to remember all the information you get during an appointment. Someone who comes with you may remember something that you missed or forgot.
- Write down questions to ask your health care team.
If you find out you have nonalcoholic fatty liver disease, some basic questions to ask include:
- Is the fat in my liver hurting my health?
- Will my fatty liver disease become serious?
- What are my treatment options?
- What can I do to keep my liver healthy?
- I have other health issues. How can I best manage them together?
- Should I see a specialist? Will my insurance cover it?
- Are there any brochures or other printed material that I can take with me? What websites do you recommend?
- Should I plan for a follow-up visit?
In addition to the questions that you've prepared to ask your care team, don't hesitate to ask questions during your appointment.
What to expect from your doctor
Your doctor is likely to ask you a number of questions, such as:
- Have you had any symptoms, such as yellowing of the eyes or skin and pain or swelling around your waist?
- If you had tests done at that time, what were the results?
- Do you drink alcohol?
- What medicines do you take, including over-the-counter medicines and supplements?
- Have you ever been told that you have hepatitis?
- Do other people in your family have liver disease?