Diagnosis
Bone marrow exam
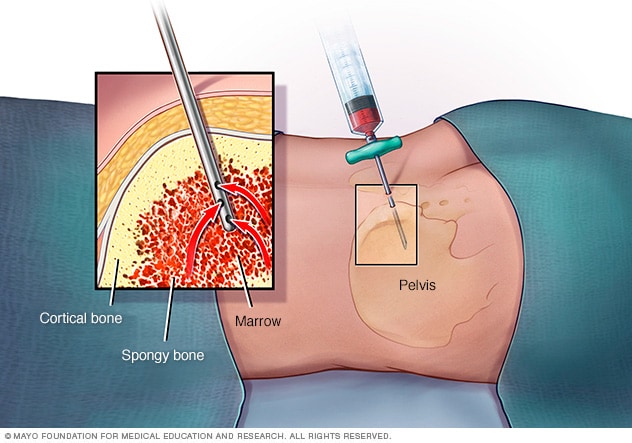
Bone marrow exam
In a bone marrow aspiration, a healthcare professional uses a thin needle to remove a small amount of liquid bone marrow. It is usually taken from a spot in the back of the hipbone, also called the pelvis. A bone marrow biopsy is often done at the same time. This second procedure removes a small piece of bone tissue and the enclosed marrow.
A physical exam, medical history and tests might be used if your doctor suspects that you have a myelodysplastic syndrome.
Tests might include:
- Blood tests. Your doctor might order blood tests to determine the number of red cells, white cells and platelets and look for unusual changes in the size, shape and appearance of various blood cells.
- Removing bone marrow for testing. During a bone marrow biopsy and aspiration, a thin needle is used to withdraw (aspirate) a small amount of liquid bone marrow, usually from a spot on the back of your hipbone. Then a small piece of bone with its marrow is removed (biopsy).
Blood and bone marrow samples are sent for laboratory analysis. Specialized tests can determine the specific characteristics of your cells that will be helpful for determining the type of myelodysplastic syndrome you have, your prognosis and your treatment options.
Treatment
Management of myelodysplastic syndromes is most often intended to slow the disease, ease symptoms and prevent complications. There's no cure for myelodysplastic syndromes, but some medications can help slow the progression of the disease.
If you have no symptoms, treatment might not be needed right away. Instead, your doctor might recommend regular exams and lab tests to monitor your condition and to see if the disease progresses.
Research on myelodysplastic syndromes is ongoing. Ask your doctor about clinical trials for which you might be eligible.
Blood transfusions
Blood transfusions with healthy blood cells from donors can be used to replace red blood cells and platelets in people with myelodysplastic syndromes. Blood transfusions can help control symptoms.
Medications
Treatment for myelodysplastic syndromes might include medications that:
- Increase the number of blood cells your body makes. Called growth factors, these medications are artificial versions of substances found naturally in your bone marrow. Growth factors that stimulate your bone marrow to make more red blood cells can help reduce your need for frequent blood transfusions. Growth factors that promote white blood cell production may reduce your risk of infection.
- Stimulate blood cells to mature. Medications that help stimulate the blood cells to mature can reduce the need for frequent blood transfusions in people who aren't helped by growth factors. Some of these drugs may also reduce the risk that the disease may progress to leukemia.
- Suppress your immune system. Medications that suppress or control your immune system are used in certain myelodysplastic syndromes to reduce your need for red blood cell transfusions.
- Help people with a certain genetic abnormality. If your myelodysplastic syndrome is associated with a gene mutation called isolated del(5q), your doctor might recommend lenalidomide (Revlimid).
- Treat infections. If your condition causes you to have infections, you'll receive treatments to control them.
Bone marrow transplant
A bone marrow transplant, also known as a stem cell transplant, is the only treatment option that offers the potential of a cure for myelodysplastic syndromes. But this treatment carries a high risk of serious complications and it's generally reserved for people who are healthy enough to endure it.
During a bone marrow transplant, high doses of chemotherapy drugs are used to clear out the defective blood cells from your bone marrow. Then the abnormal bone marrow stem cells are replaced with healthy, donated cells (allogeneic transplant).
In some situations, less intense chemotherapy drugs can be used to reduce the risks of bone marrow transplant for older adults and those who might not otherwise be considered for this treatment.
Clinical trials
Explore Mayo Clinic studies testing new treatments, interventions and tests as a means to prevent, detect, treat or manage this condition.
Lifestyle and home remedies
Because people with certain myelodysplastic syndromes have low white blood cell counts, they're subject to recurrent, and often serious, infections.
To reduce your risk of infections:
- Wash your hands. Wash hands frequently and thoroughly with warm, soapy water, especially before eating or preparing food. Carry an alcohol-based hand sanitizer for times when water isn't available.
- Take care with food. Thoroughly cook all meat and fish. Avoid fruits and vegetables you can't peel, especially lettuce, and wash all produce you do use before peeling it. To increase safety, you might want to avoid all raw foods.
- Avoid people who are ill. Try to avoid close contact with anyone who is sick, including family members and co-workers.
Preparing for your appointment
You'll likely start by seeing your family doctor or primary care doctor. If your doctor suspects that you have a myelodysplastic syndrome, you might be referred to a doctor who specializes in blood disorders (hematologist).
Here's some information to help you get ready for your appointment.
What you can do
When you make the appointment, ask if there's anything you need to do in advance, such as restrict your diet.
Make a list of:
- Your symptoms, including those that seem unrelated to the reason for the appointment, and when they began
- Key personal information, including previous treatment for cancer or exposure to toxic chemicals
- All medications, vitamins and supplements you take, including doses
- Questions to ask your doctor
Consider taking a family member or friend along to help you remember the information you're given.
For myelodysplastic syndromes, questions to ask your doctor might include:
- What type of myelodysplastic syndrome do I have?
- Will I need more tests?
- What is my prognosis?
- What is my risk of leukemia?
- If I need treatment, what are my options and what do you recommend?
- I have other health conditions. How can I best manage them together?
- Are there restrictions I need to follow?
- Are there brochures or other printed material that I can have? What websites do you recommend?
Don't hesitate to ask other questions.
What to expect from your doctor
Your doctor is likely to ask questions, such as:
- Have your symptoms been continuous or occasional?
- How severe are your symptoms?
- What, if anything, seems to improve your symptoms?
- What, if anything, appears to worsen your symptoms?
Oct. 26, 2022