Diagnosis
Bronchoscopy
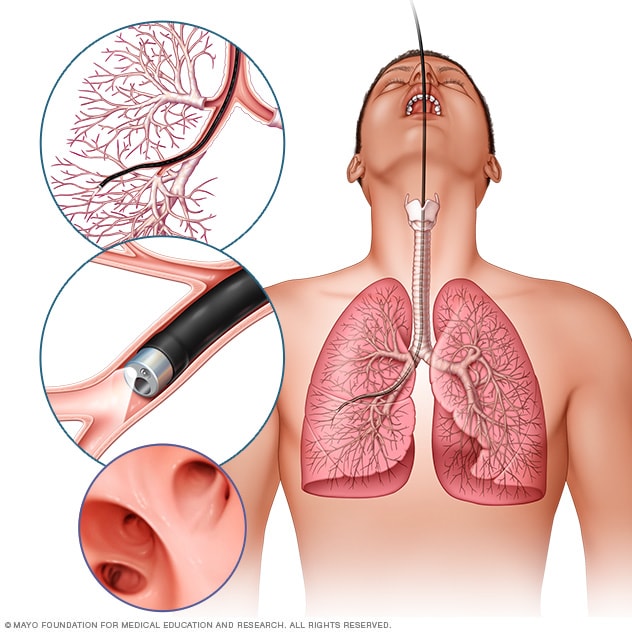
Bronchoscopy
In flexible bronchoscopy, a healthcare professional inserts a thin, bendable tube through the mouth or nose into the lungs. A light and a small camera on the bronchoscope allow the health professional to look inside the lungs' airways.
Lung cancer diagnosis often starts with an imaging test to look at the lungs. If you have symptoms that worry you, a healthcare professional might start with an X-ray. If you smoke or used to smoke, you might have an imaging test to look for signs of lung cancer before you develop symptoms.
Testing healthy people for lung cancer
People with an increased risk of lung cancer may consider yearly lung cancer screening using low-dose CT scans. Lung cancer screening is generally offered to people 50 and older who smoked heavily for many years. Screening also is offered to people who have quit smoking in the past 15 years.
Discuss your lung cancer risk with your healthcare professional. Together you can decide whether lung cancer screening is right for you.
Tests to diagnose lung cancer
If your healthcare professional thinks you may have lung cancer, a number of tests can be used to look for cancerous cells and to rule out other conditions.
Tests may include:
- Imaging tests. Imaging tests make pictures of the body. They can show the location and size of the lung cancer. Tests might include X-ray, MRI, CT and positron emission tomography, which also is called a PET scan.
- Sputum cytology. Sputum is the mucus that is coughed up from the lungs. If you are coughing up sputum, it can be looked at under a microscope. The sputum can sometimes show lung cancer cells.
-
Biopsy. A biopsy is a procedure to remove a sample of tissue for testing in a lab.
Your healthcare team can perform a lung cancer biopsy several ways. One way is bronchoscopy. During bronchoscopy, a healthcare professional passes a lighted tube with a camera down your throat into your lungs to examine the area. Special tools can be passed through the tube to collect a sample of tissue.
Mediastinoscopy also is an option. During mediastinoscopy, an incision is made at the base of your neck. Surgical tools are then inserted behind your breastbone to take tissue samples from lymph nodes.
Another option is a needle biopsy. In a needle biopsy, your healthcare professional uses X-ray or CT images to guide a needle through the skin on your chest. The needle goes into the lung tissue to collect cells that could be cancerous.
A biopsy sample also may be taken from lymph nodes or other areas where cancer has spread.
Your cancer cells will be carefully tested in a lab to find out what type of lung cancer you have. The results can help determine the likely outcome of your cancer, called the prognosis, and guide your treatment.
Tests to determine the extent of the cancer
If you're diagnosed with lung cancer, you may have other tests to see if the cancer has spread. These tests help your healthcare team find out the extent of your cancer, also called the stage. Cancer staging tests often involve imaging tests. The tests might look for signs of cancer in your lymph nodes or in other parts of your body. Your healthcare team uses the cancer staging test results to help create your treatment plan.
Imaging tests may include MRI, CT, bone scans and PET scan. Not every test is right for every person. Talk with your healthcare professional about which procedures will work for you.
The stages of lung cancer range from 1 to 4. The lowest number means that the cancer is small and only in the lung. As the cancer grows larger or spreads outside of the lungs, the numbers get higher. A stage 4 lung cancer has spread to other areas of the body.
In small cell lung cancer, the stages may be called limited or extensive. In the limited stage, the cancer affects one lung and the area around it. In the extensive stage, the cancer has spread to the other lung or to other parts of the body.
More Information
Treatment
Treatment for lung cancer usually begins with surgery to remove the cancer. If the cancer is very large or has spread to other parts of the body, surgery may not be possible. Treatment might start with medicine and radiation instead. Your healthcare team considers many factors when creating a treatment plan. These factors may include your overall health, the type and stage of your cancer, and your preferences.
Some people with lung cancer choose not to have treatment. For instance, you may feel that the side effects of treatment will outweigh the potential benefits. When that's the case, your healthcare professional may suggest comfort care to treat only the symptoms the cancer is causing.
Surgery
Lung cancer surgery
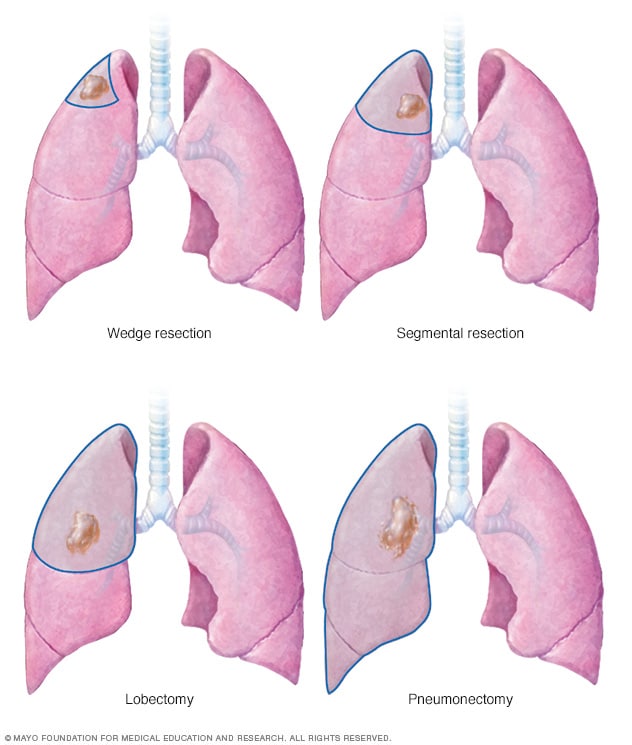
Lung cancer surgery
Lung cancer surgery can involve removing a portion of the lung or the entire lung. An operation to remove the lung cancer and a small portion of healthy tissue is called a wedge resection. Removing a larger area of the lung is called segmental resection. Surgery to remove one lobe from a lung is called lobectomy. Removing an entire lung is called pneumonectomy.
During surgery, your surgeon works to remove the lung cancer and some healthy tissue around it. Procedures to remove lung cancer include:
- Wedge resection to remove a small section of lung that contains the cancer along with a margin of healthy tissue.
- Segmental resection to remove a larger portion of lung, but not an entire lobe.
- Lobectomy to remove the entire lobe of one lung.
- Pneumonectomy to remove an entire lung.
If you have surgery, your surgeon also may remove lymph nodes from your chest to test them for cancer.
Surgery may be an option if your cancer is only in the lungs. If you have a larger lung cancer, chemotherapy or radiation therapy may be used before surgery to shrink the cancer. Chemotherapy or radiation therapy also may be used after surgery if there's a risk that cancer cells were left behind or that your cancer may come back.
Radiation therapy
Radiation therapy treats cancer with powerful energy beams. The energy can come from X-rays, protons or other sources. During radiation therapy, you lie on a table while a machine moves around you. The machine directs radiation to precise points on your body.
For lung cancer that has spread within the chest, radiation may be used before surgery or after surgery. It's often combined with chemotherapy treatments. If surgery isn't an option, combined chemotherapy and radiation therapy may be your first treatment.
For lung cancers that have spread to other areas of the body, radiation therapy may help relieve symptoms.
Chemotherapy
Chemotherapy treats cancer with strong medicines. Many chemotherapy medicines exist. Most are given through a vein. Some come in pill form. A combination of medicines usually is given in a series of treatments over a period of weeks or months. Breaks in between are used to help you recover.
Chemotherapy is often used after surgery to kill any cancer cells that may remain. It can be used alone or combined with radiation therapy. Chemotherapy also may be used before surgery to shrink cancers and make them easier to remove.
In people with lung cancer that has spread, chemotherapy can be used to relieve pain and other symptoms.
Stereotactic body radiotherapy
Stereotactic body radiotherapy is an intense radiation treatment. This treatment aims beams of radiation from many angles at the cancer. Stereotactic body radiotherapy treatment is typically completed in one or a few treatments. Sometimes this treatment is called stereotactic radiosurgery.
Stereotactic body radiotherapy may be an option for people with small lung cancers who can't have surgery. It also may be used to treat lung cancer that spreads to other parts of the body, including the brain.
Targeted therapy
Targeted therapy for cancer is a treatment that uses medicines that attack specific chemicals in the cancer cells. By blocking these chemicals, targeted treatments can cause cancer cells to die. For lung cancer, targeted therapy may be used for people with cancer that spreads or comes back after treatment.
Some targeted therapies only work in people whose cancer cells have certain DNA changes. Your cancer cells may be tested in a lab to see if these medicines might help you.
Immunotherapy
Immunotherapy for cancer is a treatment with medicine that helps the body's immune system to kill cancer cells. The immune system fights off diseases by attacking germs and other cells that shouldn't be in the body. Cancer cells survive by hiding from the immune system. Immunotherapy helps the immune system cells find and kill the cancer cells.
For lung cancer, immunotherapy might be used after surgery to kill any cancer cells that remain. When surgery isn't an option, immunotherapy might help control the cancer.
Palliative care
Palliative care is a special type of healthcare that helps you feel better when you have a serious illness. If you have cancer, palliative care can help relieve pain and other symptoms. A healthcare team that may include doctors, nurses and other specially trained health professionals provides palliative care. The care team's goal is to improve quality of life for you and your family.
Palliative care specialists work with you, your family and your care team. They provide an extra layer of support while you have cancer treatment. You can have palliative care at the same time you're getting strong cancer treatments, such as surgery, chemotherapy or radiation therapy.
The use of palliative care with other proper treatments can help people with cancer feel better and live longer.
More Information
Clinical trials
Explore Mayo Clinic studies testing new treatments, interventions and tests as a means to prevent, detect, treat or manage this condition.
Lifestyle and home remedies
Many people with lung cancer experience shortness of breath. Treatments such as supplemental oxygen and medicines are available to help you feel more comfortable. However, they aren't always enough.
To cope with shortness of breath, it may help to:
Try to relax
Feeling short of breath can be scary. But fear and anxiety only make it harder to breathe. When you begin to feel short of breath, choose an activity that helps you relax. Listen to music, imagine your favorite vacation spot, meditate or say a prayer.
Find a comfortable position
It may help to lean forward when you feel short of breath.
Focus on your breath
When you feel short of breath, focus your mind on your breathing. Instead of trying to fill your lungs with air, concentrate on moving the muscles that control your breathing. Try breathing through pursed lips and pacing your breaths with your activity.
Save your energy for what's important
If you're short of breath, you may become tired easily. Prioritize your tasks for the day so that you can save your energy for what needs to be done.
Tell your healthcare professional if you experience shortness of breath or if your symptoms worsen. There are many other treatments available to relieve shortness of breath.
Alternative medicine
Complementary and alternative lung cancer treatments can't cure your cancer. But complementary and alternative treatments can often be combined with your healthcare team's care to help relieve symptoms.
The American College of Chest Physicians suggests people with lung cancer may find comfort in:
Acupuncture
During an acupuncture session, a trained practitioner inserts small needles into precise points on your body. Acupuncture may relieve pain and ease cancer treatment side effects, such as nausea and vomiting.
Hypnosis
Hypnosis is typically done by a therapist who leads you through relaxation exercises. The therapist may ask you to think pleasing and positive thoughts. Hypnosis may reduce anxiety, nausea and pain in people with cancer.
Massage
During massages, massage therapists use their hands to apply pressure to your skin and muscles. Massage can help relieve anxiety and pain in people with cancer. Some massage therapists are specially trained to work with people who have cancer.
Meditation
Meditation is a time of quiet reflection in which you focus on something. It may be an idea, image or sound. Meditation may reduce stress and improve quality of life in people with cancer.
Yoga
Yoga combines gentle stretching movements with deep breathing and meditation. Yoga may help people with cancer sleep better.
Coping and support
With time, you'll find what helps you cope with the uncertainty and distress of a cancer diagnosis. Until then, you may find that it helps to:
Learn enough about lung cancer to make decisions about your care
Ask your healthcare team about your cancer, including your test results, treatment options and, if you like, your prognosis. As you learn more about lung cancer, you may become more confident in making treatment decisions.
Keep friends and family close
Keeping your close relationships strong will help you deal with your lung cancer. Friends and family can provide the practical support you'll need, such as helping take care of your home if you're in the hospital. And they can serve as emotional support when you feel overwhelmed by having cancer.
Find someone to talk with
Find someone who is willing to listen to you talk about your hopes and fears. This may be a friend or family member. The concern and understanding of a counselor, medical social worker, clergy member or cancer support group also may be helpful.
Ask your healthcare team about support groups in your area. Other sources of information include the National Cancer Institute and the American Cancer Society.
Preparing for your appointment
Make an appointment with a doctor or other healthcare professional if you have any symptoms that worry you.
If your healthcare professional suspects that you have lung cancer, you'll likely be referred to a specialist. Specialists who treat lung cancer may include:
- Oncologists. Doctors who specialize in treating cancer.
- Pulmonologists. Doctors who diagnose and treat lung diseases.
- Radiation oncologists. Doctors who use radiation to treat cancer.
- Thoracic surgeons. Surgeons who operate on the lungs.
- Palliative care specialists. Doctors who treat signs and symptoms of cancer and cancer treatment.
Because appointments can be brief, it's a good idea to be prepared. Here's some information to help you get ready.
What you can do
- Be aware of any pre-appointment restrictions. At the time you make the appointment, be sure to ask if there's anything you need to do in advance, such as restrict your diet.
- Write down symptoms you're experiencing, including any that may not seem related to the reason for which you scheduled the appointment.
- Write down key personal information, including major stresses or recent life changes.
- Make a list of all medicines, vitamins or supplements you're taking and the doses. Or you may prefer to bring your medicine bottles to your appointment.
- Gather your medical records. If you've had a chest X-ray or a scan done by a different healthcare professional, try to get that file and bring it to your appointment.
- Consider taking a family member or friend along. Sometimes it can be difficult to remember all the information provided during an appointment. Someone who accompanies you may remember something that you missed or forgot.
- Write down questions to ask your healthcare team.
Questions to ask if you've been diagnosed with lung cancer
Your time with your healthcare team is limited, so preparing a list of questions can help you make the most of your time together. List your questions from most important to least important in case time runs out. For lung cancer, some basic questions to ask include:
- What type of lung cancer do I have?
- May I see the chest X-ray or CT scan that shows my cancer?
- What is causing my symptoms?
- What is the stage of my lung cancer?
- Will I need more tests?
- Should my lung cancer cells be tested for gene changes that may determine my treatment options?
- Has my cancer spread to other parts of my body?
- What are my treatment options?
- Will any of these treatment options cure my cancer?
- What are the potential side effects of each treatment?
- Is there one treatment that you think is best for me?
- Is there a benefit if I quit smoking now?
- What advice would you give a friend or family member in my situation?
- What if I don't want treatment?
- Are there ways to relieve the symptoms I'm experiencing?
- Can I enroll in a clinical trial?
- Should I see a specialist? What will that cost, and will my insurance cover it?
- Are there brochures or other material that I can take with me? What websites do you recommend?
Don't hesitate to ask other questions.
What to expect from your doctor
Be prepared to answer questions, such as:
- When did you first begin experiencing symptoms?
- Have your symptoms been ongoing or occasional?
- How severe are your symptoms?
- Do you wheeze when breathing?
- Do you have a cough that feels like you're clearing your throat?
- Have you ever been diagnosed with emphysema or chronic obstructive pulmonary disease?
- Do you take medicines for shortness of breath?
- What, if anything, seems to improve your symptoms?
- What, if anything, appears to worsen your symptoms?