Diagnosis
Endoscopic retrograde cholangiopancreatography

Endoscopic retrograde cholangiopancreatography
Endoscopic retrograde cholangiopancreatography (ERCP) uses a dye to highlight the bile ducts and pancreatic duct on X-ray images. A thin, flexible tube with a camera on the end, called an endoscope, is passed down your throat and into your small intestine. The dye enters the ducts through a small hollow tube, called a catheter, passed through the endoscope. Tiny tools passed through the catheter also can be used to remove gallstones.
Endoscopic ultrasound
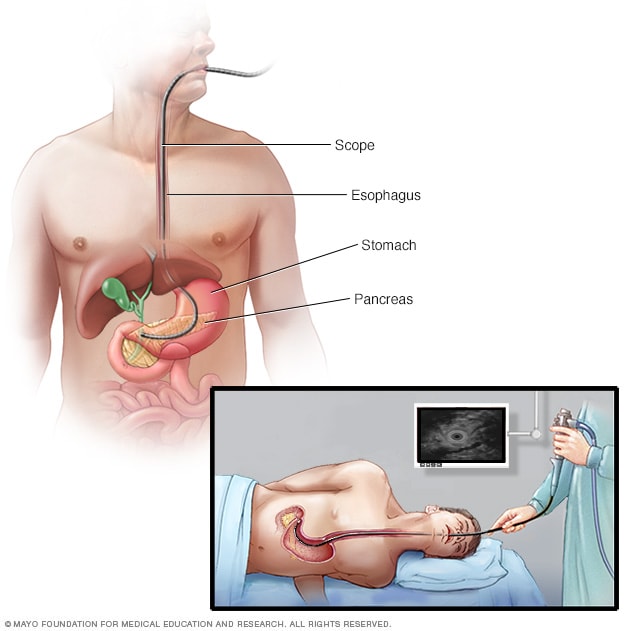
Endoscopic ultrasound
During an endoscopic ultrasound, your doctor inserts a long, flexible tube (endoscope) down your throat and into your abdomen. An ultrasound device at the end of the tube emits sound waves that generate images of nearby tissues.
If your doctor suspects cholangiocarcinoma, he or she may have you undergo one or more of the following tests:
- Liver function tests. Blood tests to measure your liver function can give your doctor clues about what's causing your signs and symptoms.
Tumor marker test. Checking the level of carbohydrate antigen (CA) 19-9 in your blood may give your doctor additional clues about your diagnosis. CA 19-9 is a protein that's overproduced by bile duct cancer cells.
A high level of CA 19-9 in your blood doesn't mean you have bile duct cancer, though. This result can also occur in other bile duct diseases, such as bile duct inflammation and obstruction.
- A test to examine your bile duct with a small camera. During endoscopic retrograde cholangiopancreatography (ERCP), a thin, flexible tube equipped with a tiny camera is passed down your throat and through your digestive tract to your small intestine. The camera is used to examine the area where your bile ducts connect to your small intestine. Your doctor may also use this procedure to inject dye into the bile ducts to help them show up better on imaging tests.
- Imaging tests. Imaging tests can help your doctor see your internal organs and look for signs of cholangiocarcinoma. Techniques used to diagnose bile duct cancer include ultrasound, computerized tomography (CT) scans and magnetic resonance imaging (MRI) combined with magnetic resonance cholangiopancreatography (MRCP). MRCP is increasingly being used as a noninvasive alternative to ERCP. It offers 3D images without the need for a dye to enhance the images.
A procedure to remove a sample of tissue for testing. A biopsy is a procedure to remove a small sample of tissue for examination under a microscope.
If the suspicious area is located very near where the bile duct joins the small intestine, your doctor may obtain a biopsy sample during ERCP. If the suspicious area is within or near the liver, your doctor may obtain a tissue sample by inserting a long needle through your skin to the affected area (fine-needle aspiration). He or she may use an imaging test, such as an endoscopic ultrasound or CT scan, to guide the needle to the precise area.
How your doctor collects a biopsy sample may influence which treatment options are available to you later. For example, if your bile duct cancer is biopsied by fine-needle aspiration, you will become ineligible for liver transplantation. Don't hesitate to ask about your doctor's experience with diagnosing cholangiocarcinoma. If you have any doubts, get a second opinion.
If your doctor confirms a diagnosis of cholangiocarcinoma, he or she tries to determine the extent (stage) of the cancer. Often this involves additional imaging tests. Your cancer's stage helps determine your prognosis and your treatment options.
More Information
Treatment
Treatments for cholangiocarcinoma (bile duct cancer) may include:
- Surgery. When possible, surgeons try to remove as much of the cancer as they can. For very small bile duct cancers, this involves removing part of the bile duct and joining the cut ends. For more-advanced bile duct cancers, nearby liver tissue, pancreas tissue or lymph nodes may be removed as well.
- Liver transplant. Surgery to remove your liver and replace it with one from a donor (liver transplant) may be an option in certain situations for people with hilar cholangiocarcinoma. For many, a liver transplant can be a cure for hilar cholangiocarcinoma, but there is a risk that the cancer will recur after a liver transplant.
- Chemotherapy. Chemotherapy uses drugs to kill cancer cells. Chemotherapy may be used before a liver transplant. It may also be an option for people with advanced cholangiocarcinoma to help slow the disease and relieve signs and symptoms. Chemotherapy drugs can be infused into a vein so that they travel throughout the body. Or the drugs can be administered in a way so that they are delivered directly to the cancer cells.
- Radiation therapy. Radiation therapy uses high-powered energy beams from sources such as X-rays and protons to kill cancer cells. Radiation therapy can involve a machine that directs radiation beams at your body (external beam radiation). Or it can involve placing radioactive material inside your body near the site of your cancer (brachytherapy).
- Targeted drug therapy. Targeted drug treatments focus on specific abnormalities present within cancer cells. By blocking these abnormalities, targeted drug treatments can cause cancer cells to die. Your doctor may test your cancer cells to see if targeted therapy may be effective against your cholangiocarcinoma.
- Immunotherapy. Immunotherapy uses your immune system to fight cancer. Your body's disease-fighting immune system may not attack your cancer because the cancer cells produce proteins that help them hide from the immune system cells. Immunotherapy works by interfering with that process. For cholangiocarcinoma, immunotherapy might be an option for advanced cancer when other treatments haven't helped.
- Heating cancer cells. Radiofrequency ablation uses electric current to heat and destroy cancer cells. Using an imaging test as a guide, such as ultrasound, the doctor inserts one or more thin needles into small incisions in your abdomen. When the needles reach the cancer, they're heated with an electric current, destroying the cancer cells.
- Photodynamic therapy. In photodynamic therapy, a light-sensitive chemical is injected into a vein and accumulates in the fast-growing cancer cells. Laser light directed at the cancer causes a chemical reaction in the cancer cells, killing them. You'll typically need multiple treatments. Photodynamic therapy can help relieve your signs and symptoms, and it may also slow cancer growth. You'll need to avoid sun exposure after treatments.
- Biliary drainage. Biliary drainage is a procedure to restore the flow of bile. It might involve placing a thin tube into the bile duct in order to drain the bile. Other strategies include bypass surgery to reroute the bile around the cancer and stents to hold open a bile duct being collapsed by cancer. Biliary drainage helps relieve signs and symptoms of cholangiocarcinoma.
Because cholangiocarcinoma is a very difficult type of cancer to treat, don't hesitate to ask about your doctor's experience with treating the condition. If you have any doubts, get a second opinion.
Clinical trials
Clinical trials are studies to test new treatments, such as new drugs and new approaches to surgery. If the treatment being studied proves to be safer and more effective than are current treatments, it can become the new standard of care.
Clinical trials can't guarantee a cure, and they might have serious or unexpected side effects. On the other hand, cancer clinical trials are closely monitored to ensure they're conducted as safely as possible. They offer access to treatments that wouldn't otherwise be available to you.
Talk to your doctor about what clinical trials might be appropriate for you.
Supportive (palliative) care
Palliative care is specialized medical care that focuses on providing relief from pain and other symptoms of a serious illness. Palliative care specialists work with you, your family and your other doctors to provide an extra layer of support that complements your ongoing care. Palliative care can be used while undergoing aggressive treatments, such as surgery.
When palliative care is used along with other appropriate treatments — even soon after your diagnosis — people with cancer may feel better and may live longer.
Palliative care is provided by teams of doctors, nurses and other specially trained professionals. These teams aim to improve the quality of life for people with cancer and their families. Palliative care is not the same as hospice care or end-of-life care.
More Information
Clinical trials
Explore Mayo Clinic studies testing new treatments, interventions and tests as a means to prevent, detect, treat or manage this condition.
Coping and support
Learning you have a life-threatening illness can be devastating. You will not find any easy answers for dealing with cholangiocarcinoma, but some of the following suggestions may help:
- Learn what you need to know about your cancer. Ask your doctor about your cancer, including the type and stage of your cancer, your treatment options and, if you like, your prognosis. As you learn more about cholangiocarcinoma, you may become more confident in making treatment decisions. Ask about trusted sources of further information.
- Keep friends and family close. Keeping your close relationships strong will help you deal with your cancer. Friends and family can provide the practical support you'll need, such as helping take care of your home if you're in the hospital. And they can serve as emotional support when you feel overwhelmed.
- Find someone to talk with. Although friends and family can be your best allies, in some cases they have difficulty coping with the shock of your diagnosis. In these cases, talking with a counselor, medical social worker, or a pastoral or religious counselor can be helpful. Ask your doctor for a referral.
- Connect with other cancer survivors. You may find comfort in talking with other cancer survivors. Contact your local chapter of the American Cancer Society to find cancer support groups in your area.
Make plans for the unknown. Having a life-threatening illness, such as cancer, requires you to prepare for the possibility that you may die. For some people, having a strong faith or a sense of something greater than themselves makes it easier to come to terms with a life-threatening illness.
Ask your doctor about advance directives and living wills to help you plan for end-of-life care, should you need it.
Preparing for your appointment
Start by making an appointment with your doctor if you have any signs or symptoms that worry you. If you doctor determines that you have cholangiocarcinoma, he or she may refer you to a doctor who specializes in diseases of the digestive system (gastroenterologist) or to a doctor who specializes in treating cancer (oncologist).
What you can do
- Be aware of any pre-appointment restrictions, such as restricting your diet.
- List your symptoms, including any that seem unrelated to the reason you scheduled the appointment.
- List key personal information, including recent changes or stressors.
- List all of your medications, vitamins and supplements, including doses.
- Ask a relative or friend to accompany you, to help you remember what the doctor says.
- List questions to ask your doctor.
- Bring copies of your medical records to your appointment, if you're seeing a new doctor for the first time. If you've had scans done at another facility, ask that files containing those images be placed on a CD and bring that to your appointment.
Some basic questions to ask your doctor include:
- Do I have bile duct cancer? What type?
- What is the stage of my cancer?
- What does my pathology report say? Can I have a copy of the pathology report?
- Will I need more tests?
- What are my treatment options?
- What are the potential side effects of each treatment option?
- Is there one treatment you think is best for me?
- How will my treatment affect my daily life?
- How much time can I take to make my decision about bile duct cancer treatment?
- What is your experience with bile duct cancer diagnosis and treatment? How many surgical procedures for this type of cancer are done each year at this medical center?
- Should I see a bile duct cancer specialist? What will that cost, and will my insurance cover it?
- Do you have any brochures or other printed material that I can take with me? What websites do you recommend?
In addition to the questions that you've prepared to ask your doctor, don't hesitate to ask additional questions during your appointment.
What to expect from your doctor
Your doctor is likely to ask you a number of questions, such as:
- When did you first begin experiencing symptoms?
- How severe are your symptoms? Are they occasional or continuous?
- Does anything improve or worsen your symptoms?
Share your experience
Share your Mayo Clinic experience with others using social media.