Diagnosis
Cervical cancer FAQs
Get answers to the most frequently asked questions about cervical cancer from Mayo Clinic gynecologic oncologist Kristina Butler, M.D., M.S.
Hello. I'm Dr. Kristina Butler, a gynecologic oncologist at Mayo Clinic, and I'm here to answer some of the important questions you might have about cervical cancer.
What is the right amount of time between Pap smears?
Pap smear screenings begin at age 21 and continue, varying by age, every three to five years.
What does it mean to have an abnormal Pap smear?
Having an abnormal Pap smear is very common, so don't feel alone. It just means that additional tests are needed to prove that cervical cancer isn't present. Most people with abnormal Pap smear do not end up having a diagnosis of cervical cancer.
What is the age cut off for the HPV vaccine?
Most recently, the HPV vaccination has been increased to include all adults male and female to age 45.
Should I still get the HPV vaccine if I already have cervical pre-cancer?
Absolutely. After cervical pre-cancer or even HPV exposure, the vaccine remains to provide benefit. We have good evidence to support this. So I recommend HPV vaccination after pre-cancer or even after cervical cancer.
Does catching cervical cancer early make a difference?
Certainly, we aim to detect cervical cancer as early as possible. Early stage cervical cancer has much improved overall survival and reduced recurrence.
Does cervical cancer impact fertility?
Even though you've been diagnosed with cervical cancer, family planning is still possible. When diagnosed at early stages, we have fertility sparing treatment options that allow a woman to be treated for cervical cancer in some cases and continue to have fertility.
How can I be the best partner to my medical team?
I highly recommend having a medical team that you're comfortable communicating with. It's always safe to ask questions, get second opinions. And remember, at the end of the day, we're all here to help you on the same team. Never hesitate to ask your medical team any questions or concerns you have. Being informed makes all the difference. Thanks for your time and we wish you well.
Screening
Pap test
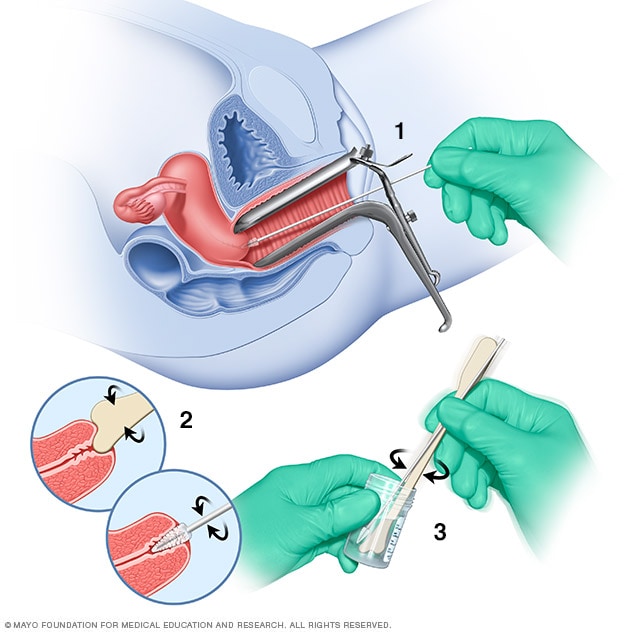
Pap test
During a Pap test, a tool called a speculum holds the vaginal walls apart. A sample of cells from the cervix is collected using a soft brush and a flat scraping device called a spatula (1 and 2). The cells are placed in a bottle that contains a solution to preserve them (3). Or the cells may be smeared onto a glass slide. Later, the cells are checked under a microscope.
Screening tests can help detect cervical cancer and precancerous cells that may one day develop into cervical cancer. Most medical organizations suggest beginning screening for cervical cancer and precancerous changes at age 21. The tests are usually repeated every few years.
Screening tests include:
-
Pap test. During a Pap test, a member of your health care team scrapes and brushes cells from your cervix. The cells are then examined in a lab to check for cells that look different.
A Pap test can detect cancer cells in the cervix. It also can detect cells that have changes that increase the risk of cervical cancer. These are sometimes called precancerous cells.
- HPV DNA test. The HPV DNA test involves testing cells from the cervix for infection with any of the types of HPV that are most likely to lead to cervical cancer.
Discuss your cervical cancer screening options with your health care team.
Diagnosis
Cone biopsy
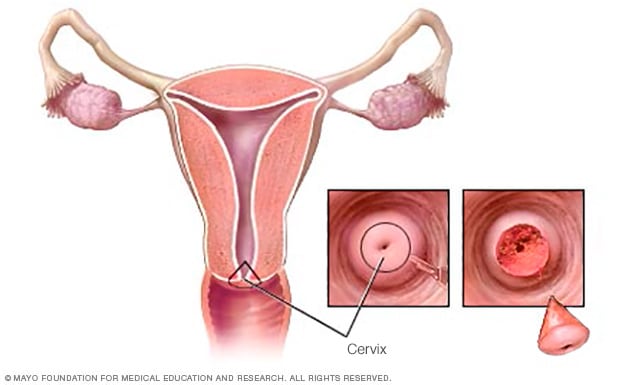
Cone biopsy
During a cone biopsy, also called conization, a doctor surgically removes a cone-shaped piece of tissue from the cervix. Typically, the cone-shaped piece includes tissue from both the upper and lower parts of the cervix.
If you might have cervical cancer, testing is likely to start with a thorough exam of your cervix. A special magnifying instrument, called a colposcope, is used to check for signs of cancer.
During the colposcopic exam, a doctor removes a sample of cervical cells for lab testing. To get the sample, you might need:
- Punch biopsy, which uses a sharp tool to pinch off small samples of cervical tissue.
- Endocervical curettage, which uses a small, spoon-shaped instrument, called a curet, or a thin brush to scrape a tissue sample from the cervix.
If the results of these tests are concerning, you might have more tests. These might include:
- Electrical wire loop, which uses a thin, low-voltage electrified wire to take a small tissue sample. Generally, this is done in a doctor's office. You receive medicine to numb the area to lessen any discomfort during the procedure. This test also may be called a loop electrosurgical excision procedure, also known as LEEP.
- Cone biopsy, also called conization, is a procedure that allows your doctor to take deeper layers of cervical cells for testing. A cone biopsy is often done in a hospital. You may receive medicine to put you in a sleep-like state so that you won't be aware during the procedure.
Staging
If you're diagnosed with cervical cancer, you might need other tests to find out the extent of the cancer, also called the stage. Your health care team uses the information from staging tests to plan your treatment.
Tests used for cervical cancer staging include:
- Imaging tests. Imaging tests make pictures of the body. They can show the location and size of the cancer. Tests might include X-ray, MRI, CT and positron emission tomography (PET) scan.
- Visual examination of your bladder and rectum. Your doctor may use special scopes to look for signs of cancer inside your bladder and rectum.
The stages of vaginal cancer range from 1 to 4. The lowest number means that the cancer is only in the cervix. As the numbers get higher, the cancer is more advanced. A stage 4 cervical cancer may have grown to involve nearby organs or spread to other areas of the body.
More Information
Treatment
Treatment for cervical cancer depends on several factors, such as the stage of the cancer, other health conditions you may have and your preferences. Surgery, radiation, chemotherapy or a combination of the three may be used.
Surgery
Small cervical cancers that haven't grown beyond the cervix are typically treated with surgery. The size of your cancer, its stage and whether you would like to consider becoming pregnant in the future will determine which operation is best for you.
Options might include:
- Surgery to cut away the cancer only. For a very small cervical cancer, it might be possible to remove all the cancer with a cone biopsy. This procedure involves cutting away a cone-shaped piece of cervical tissue and leaving the rest of the cervix intact. This option may make it possible for you to consider becoming pregnant in the future.
- Surgery to remove the cervix, called a trachelectomy. A small cervical cancer might be treated with a radical trachelectomy procedure. This procedure removes the cervix and some surrounding tissue. The uterus remains after this procedure, so it may be possible to become pregnant, if you choose.
- Surgery to remove the cervix and uterus, called a hysterectomy. Most cervical cancers that have not spread beyond the cervix are treated with a radical hysterectomy operation. This involves removing the cervix, uterus, part of the vagina and nearby lymph nodes. A hysterectomy can often cure the cancer and stop it from coming back. But removing the uterus makes it impossible to become pregnant.
Minimally invasive hysterectomy may be an option for very small cervical cancers that have not spread, known as microinvasive cancers. This procedure involves making several small cuts in the abdomen rather than one large cut. People who have minimally invasive surgery tend to recover faster and spend less time in the hospital. But some research has found that minimally invasive hysterectomy may be less effective than traditional hysterectomy. If you're considering minimally invasive surgery, discuss the benefits and risks of this approach with your surgeon.
Radiation therapy
Radiation therapy uses powerful energy beams to kill cancer cells. The energy can come from X-rays, protons or other sources. Radiation therapy is often combined with chemotherapy as the primary treatment for cervical cancers that have grown beyond the cervix. It also can be used after surgery if there's an increased risk that the cancer will come back.
Radiation therapy can be given:
- Externally, called external beam radiation therapy. A radiation beam is directed at the affected area of the body.
- Internally, called brachytherapy. A device filled with radioactive material is placed inside your vagina, usually for only a few minutes.
- Both externally and internally.
If you haven't started menopause, radiation therapy might cause menopause. Ask your health care team about ways to preserve your eggs before treatment.
Chemotherapy
Chemotherapy uses strong medicines to kill cancer cells. For cervical cancer that has spread beyond the cervix, low doses of chemotherapy are often combined with radiation therapy. This is because chemotherapy may enhance the effects of the radiation. Higher doses of chemotherapy might be recommended to help control symptoms of very advanced cancer. Chemotherapy may be used before surgery to reduce the size of the cancer.
Targeted therapy
Targeted therapy uses medicines that attack specific chemicals in the cancer cells. By blocking these chemicals, targeted treatments can cause cancer cells to die. Targeted therapy is usually combined with chemotherapy. It might be an option for advanced cervical cancer.
Immunotherapy
Immunotherapy is a treatment with medicine that helps your immune system kill cancer cells. Your immune system fights off diseases by attacking germs and other cells that shouldn't be in your body. Cancer cells survive by hiding from the immune system. Immunotherapy helps the immune system cells find and kill the cancer cells. For cervical cancer, immunotherapy might be considered when the cancer is advanced and other treatments aren't working.
Palliative care
Palliative care is a special type of health care that helps you feel better when you have a serious illness. If you have cancer, palliative care can help relieve pain and other symptoms. A team that can include doctors, nurses and other specially trained professionals provides palliative care. The team's goal is to improve quality of life for you and your family.
Palliative care specialists work with you, your family and your care team to help you feel better. They provide an extra layer of support while you have cancer treatment. You can have palliative care at the same time as strong cancer treatments, such as surgery, chemotherapy or radiation therapy.
Using palliative care along with all the other appropriate treatments can help people with cancer feel better and live longer.
More Information
Clinical trials
Explore Mayo Clinic studies testing new treatments, interventions and tests as a means to prevent, detect, treat or manage this condition.
Coping and support
With time, you'll find what helps you cope with the uncertainty and distress of a cancer diagnosis. Until then, you may find that it helps to:
- Learn enough about cervical cancer to make decisions about your care. Write down your questions for your health care team and ask them at the next appointment. Get a friend or family member to come to appointments with you to take notes. Ask your health care team for further sources of information.
- Find someone to talk with. You may feel comfortable discussing your feelings with a friend or family member, or you might prefer meeting with a formal support group. Support groups for the families of cancer survivors also are available.
- Let people help. Cancer treatments can be tiring. Let friends and family know what types of help would be most useful for you.
- Set reasonable goals. Having goals helps you feel in control and can give you a sense of purpose. But choose goals that you can reach.
- Take time for yourself. Eating well, relaxing and getting enough rest can help combat the stress and fatigue of cancer.
Preparing for your appointment
Make an appointment with a doctor or other health care professional if you have symptoms that worry you. If a health professional thinks you might have cervical cancer, you may be referred to a doctor who specializes in treating cancers that affect the female reproductive system, called a gynecologic oncologist.
Here's some information to help you get ready for your appointment and what to expect from your health care team.
What you can do
- Be aware of any pre-appointment restrictions, such as not eating solid food on the day before your appointment.
- Write down your symptoms, including any that seem to not be related to the reason why you scheduled the appointment.
- Write down your key medical information, including other conditions.
- Write down key personal information, including anything that increases your risk of STIs, such as early sexual activity, multiple partners or unprotected sex.
- Make a list of all your medicines, vitamins or supplements.
- Ask a relative or friend to come with you, to help you remember what your health care team says.
- Write down questions to ask your team.
Questions to ask your doctor
- What's the most likely cause of my symptoms?
- What kinds of tests do I need?
- What treatments are available, and what side effects can I expect?
- What is the prognosis?
- How often will I need follow-up visits after I finish treatment?
In addition to the questions that you've prepared, don't hesitate to ask other questions that occur to you.
What to expect from your doctor
Be prepared to answer some questions about your symptoms and your health history, such as:
- What symptoms are you experiencing? How severe are they?
- When did you first begin experiencing symptoms? Have they changed over time?
- Have you had regular Pap tests since you became sexually active? Have you ever had irregular Pap test results in the past?
- Have you ever been treated for a cervical condition?
- Have you ever been diagnosed with an STI?
- Have you ever taken medications that suppress your immune system?
- Do you or have you ever smoked? How much?
- Do you want to have children in the future?
Sept. 02, 2023